Products Categories
| CAS No.: | 12037-29-5 |
|---|---|
| Name: | Praseodymium oxide |
| Molecular Structure: | |
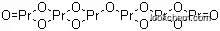 |
|
| Formula: | O11Pr6 |
| Molecular Weight: | 1021.44 |
| Synonyms: | Hexapraseodymiumundecaoxide;Praseodymium oxide (Pr12O22);Praseodymium undecaoxide; |
| EINECS: | 234-857-9 |
| Density: | 6.5g/mLat 25°C(lit.) |
| Melting Point: | 2500°C |
| Solubility: | Insoluble in water |
| Appearance: | Black powder |
| Hazard Symbols: |
 Xi Xi
|
| Risk Codes: | 36/37/38 |
| Safety: | 26-37/39 |
| PSA: | 0.00000 |
| LogP: | -0.35640 |
- 144851-82-1METHYL2-AMINO-3-FLUOROBENZOATE
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 1219080-61-1IMIDAZOLE-2-BORONIC ACID
Specification
The Praseodymium oxide(Pr6O11), with the cas registry number 12037-29-5, has the IUPAC name of oxygen(2-); praseodymium(3+). This is a kind of dark green to dark brown or black powder and is insoluble in water.
The product categories of this chemical are as follows: Inorganics; Rare earth; PraseodymiumMetal and Ceramic Science; Catalysis and Inorganic Chemistry; Chemical Synthesis; Oxides; Praseodymium; 59: Pr; PraseodymiumNanomaterials; Materials Science; Nanomaterials; Nanoparticles: Oxides, Nitrides, and Other CeramicsChemical Synthesis; Nanopowders and Nanoparticle Dispersions
The characteristics of this chemical are as follows: (1)#H bond acceptors: 1 ; (2)#H bond donors: 0 ; (3)Polar Surface Area: 0; (4)Exact Mass: 1021.389949; (5)MonoIsotopic Mass: 1021.389949; (6)Topological Polar Surface Area: 11; (7)Heavy Atom Count: 17; (8)Formal Charge: -4.
The production method of this chemical is below: monazite concentrates go through the alkali solution, phosphorus moving out, leaching of diluted hydrochloric acid to get the rare earth chloride solvent; Next extract with P2O4-kerosene-HCl-ReCl system to divide it into three group which are light, middle, heavy rare earth; Then raffinate La-Nd light rare earth will be divided according to the oxalate sediment sequence which is samarium-neodymium (Nd)-praseodymium-cerium-lanthanum; Following, oxalate sediment could produce soluble complex and then seperate, dry to get the Praseodymium oxide(Pr6O11) at 1000℃. The chemical equation is as below: 3Pr2(C2O4)3→Pr6O11+7CO2+11CO.
As to its usage, it is widely applied in many ways. When used in ceramics, it could be the material of making praseodymium yellow; When used in glass industry, it could be as the coloring agent and could also copy the gemstone; If applied in metallurgical industry, it could be the material of producing praseodymium, samarium, cobalt (Co) and other permanent magnet alloy.
When you are dealing with this chemical, you should be cautious. This is irritating to eyes, respiratory system and skin and may may cause inflammation to the skin or other mucous membranes. Therefore, you should wear suitable gloves and eye/face protection and if in case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
Additionally, you could convert the following datas into the molecular structure:
(1)Canonical SMILES: [O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[Pr+3]
.[Pr+3].[Pr+3].[Pr+3].[Pr+3].[Pr+3]
(2)InChI: InChI=1S/11O.6Pr/q11*-2;6*+3
(3)InChIKey: HPZIIFVSYNLWNX-UHFFFAOYSA-N

