Products Categories
| CAS No.: | 12069-32-8 |
|---|---|
| Name: | Boron carbide |
| Molecular Structure: | |
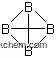 |
|
| Formula: | CB4 |
| Molecular Weight: | 55.25 |
| Synonyms: | B 4C-C1005;Boroncarbide;Boron carbide (B0.8C0.2);Boron carbide (B12C3);Ceralloy 546-3E;DB1031J;Denkaboron 1200;Denkaboron 1500;Denkaboron F 1;ESK 1500;F 1200;F1500;HS (carbide);Norbide;Stark HS;Tetrabor 1000;Tetrabor1500;Tetrabor 500;Tetraboron carbide;Tetraboron monocarbide; |
| EINECS: | 235-111-5 |
| Density: | 2.51 g/cm3 |
| Melting Point: | 2450°C |
| Boiling Point: | 3500°C |
| Solubility: | Insoluble in water. |
| Appearance: | off-white solid |
| Hazard Symbols: |
 Xn Xn
|
| Risk Codes: | 20 |
| Safety: | 22-36/37/39-38 |
| PSA: | 0.00000 |
| LogP: | -2.87230 |
- 144851-82-1METHYL2-AMINO-3-FLUOROBENZOATE
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 1219080-61-1IMIDAZOLE-2-BORONIC ACID
Specification
The Boron carbide, with its cas registry number 12069-32-8, has the systematic name of 2,3,4,5-tetraboratetracyclo[2.1.0.0~1,3~.0~2,5~]pentane. This is a kind of black odourless powder, and is stable chemically while is incompatible with oxidizing agents. Besides, it belongs to the product categories which are inorganics.
The characteristics of this chemical are as follows: (1)Index of Refraction: 1.347; (2)Molar Refractivity: 12.39 cm3; (3)Molar Volume: 58 cm3; (4)Polarizability: 4.91× 10-24 cm3; (5)Surface Tension: 4.5 dyne/cm; (6)Density: 0.95 g/cm3; (7)Enthalpy of Vaporization: 17.24 kJ/mol; (8)Vapour Pressure: 24600 mmHg at 25°C; (9)Exact Mass: 56.037222; (10)MonoIsotopic Mass: 56.037222; (11)Topological Polar Surface Area: 0; (12)Heavy Atom Count: 5; (13)Complexity: 106.
The production method of this chemical is below: smash the graphite and coal and then put into the ball mill with the boric acid with the charge ratio of 0.5:0.5:3 to have the complete blending; Next keep them in the arc furnace to have the carbonization reaction at 1700~2300℃; Then go through the process of ejection, cooling, smashing, choosing, wahsing and then coarse crushing, grinding, scouring for 12 hours at no less than 80℃; Finally wash to be neutral, then sorting with the sediment method and series washing, and then dry to get the products. The chemical equation of this way is below: 4H3BO3+7C→B4C+6CO↑+6H2O.
As to its usage, it is widely applied in many ways. It could be used as the grinding material, wear-resistant material in mechanograph and then also in nuclear reactors; It could be also used in the drilling and polishing for the stereoplasm materials and also in production of metal boride.
When you are using this chemical, please be careful. For being a kind of harmful chemical, it may cause damage to health. If by inhalation, it will be very dangerous. While using, you had better take the following advices. Wear suitable protective clothing, gloves and eye/face protection and if in case of insufficient ventilation, wear suitable respiratory equipment. Then remember not to breathe dust.
Additionally, you could convert the following datas into the molecular structure:
(1)Canonical SMILES: B12B3C14B2B43
(2)InChI: InChI=1S/CB4/c2-1-3(2)5(1)4(1)2
(3)InChIKey: INAHAJYZKVIDIZ-UHFFFAOYSA-N

