Products Categories
| CAS No.: | 125-10-0 |
|---|---|
| Name: | Prednisone 21-acetate |
| Article Data: | 40 |
| Molecular Structure: | |
|
|
|
| Formula: | C23H28O6 |
| Molecular Weight: | 400.472 |
| Synonyms: | Pregna-1,4-diene-3,11,20-trione,17,21-dihydroxy-, 21-acetate (6CI,7CI,8CI);21-Acetoxy-17a-hydroxypregna-1,4-diene-3,11,20-trione;Cortancyl;Delcortin;Delta-Corlin;Deltalone;Ferrosan;NSC 10965;Nisone;Prednisone acetate;D1-Cortisone 21-acetate;D1-Dehydrocortisone acetate; |
| EINECS: | 204-726-0 |
| Density: | 1.28 g/cm3 |
| Melting Point: | 240-242°C (dec.) |
| Boiling Point: | 582 °C at 760 mmHg |
| Flash Point: | 200.2 °C |
| Solubility: | 23mg/L(25 oC) |
| Hazard Symbols: |
 Xi Xi
|
| Risk Codes: | R20/21/22; R36/37/38 |
| PSA: | 97.74000 |
| LogP: | 2.33660 |
- 144851-82-1METHYL2-AMINO-3-FLUOROBENZOATE
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE

| Conditions | Yield |
|---|---|
| With 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione In N,N-dimethyl-formamide at 23 - 25℃; for 5h; | 99% |
| With 1-hydroxy-1.oxo-1H-1λ5-benzo[d][1,2]iodoxol-3-one pyridinium salt In N,N-dimethyl-formamide at 24 - 28℃; for 4h; | 97% |
| With N-bromoacetamide | |
| With chromium(VI) oxide |

| Conditions | Yield |
|---|---|
| With tert-Butyl peroxybenzoate Inert atmosphere; Reflux; | 81% |

| Conditions | Yield |
|---|---|
| With selenium(IV) oxide | |
| With periodic acid | |
| With iodine pentoxide | |
| With Arthrobacter simplex By-2-13 | 17 g |
- 52-21-1
prednisolone 21-acetate

A
- 898-84-0
(8S,9S,10R,11S,13S,14S)-11-Hydroxy-10,13-dimethyl-7,8,9,10,11,12,13,14,15,16-decahydro-6H-cyclopenta[a]phenanthrene-3,17-dione

B
- 125-10-0
prednisone acetate

| Conditions | Yield |
|---|---|
| Product distribution; Rate constant; Irradiation; radiolytic degradation; |

- 125-10-0
prednisone acetate

| Conditions | Yield |
|---|---|
| With selenium(IV) oxide | |
| With iodine pentoxide; periodic acid |

- 125-10-0
prednisone acetate

| Conditions | Yield |
|---|---|
| With selenium(IV) oxide | |
| With iodine pentoxide; periodic acid | |
| Bromierung und Erhitzen mit 2,4,6-Trimethyl-pyridin; |

| Conditions | Yield |
|---|---|
| In pyridine |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: pyridine 2: CrO3 View Scheme |
- 28449-43-6
21-acetoxy-11β-hydroxy-pregna-1,4,17(20)c-trien-3-one

- 125-10-0
prednisone acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: diacetoxyiodanyl-benzene 2: N-bromo-acetamide View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: acetic acid; manganese(ll) chloride; chromium(VI) oxide / water; chloroform / 30 °C 2: Arthrobacter simplex By-2-13 View Scheme |

What can I do for you?
Get Best Price
Specification
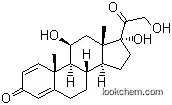
The Prednisone 21-acetate, with the CAS registry number 125-10-0, is also known as Prednisone acetate. It belongs to the product categories of Hormone; Biochemical Reagents; Biochemicals. Its EINECS registry number is 204-726-0. This chemical's molecular formula is C23H28O6 and molecular weight is 400.46482. Its IUPAC name is called [2-[(8S,9S,10R,13S,14S,17R)-17-hydroxy-10,13-dimethyl-3,11-dioxo-6,7,8,9,12,14,15,16-octahydrocyclopenta[a]phenanthren-17-yl]-2-oxoethyl]acetate. This chemical's classification codes are Hormone; Reproductive Effect. It is a kind of adrenal cortical hormone drug and has capabilities of anti-inflammatory, antiallergic. Besides that it is also used for biochemical studies. In addition, it is obtained from acetic acid cortisone via dehydrogenation of selenium dioxide.
Physical properties of Prednisone 21-acetate: (1)ACD/LogP: 2.66; (2)ACD/LogD (pH 5.5): 2.66; (3)ACD/LogD (pH 7.4): 2.66; (4)ACD/BCF (pH 5.5): 61.51; (5)ACD/BCF (pH 7.4): 61.51; (6)ACD/KOC (pH 5.5): 663.95; (7)ACD/KOC (pH 7.4): 663.94; (8)#H bond acceptors: 6; (9)#H bond donors: 1; (10)#Freely Rotating Bonds: 5; (11)Index of Refraction: 1.579; (12)Molar Refractivity: 103.66 cm3; (13)Molar Volume: 311.5 cm3; (14)Surface Tension: 54.3 dyne/cm; (15)Density: 1.28 g/cm3; (16)Flash Point: 200.2 °C; (17)Enthalpy of Vaporization: 99.92 kJ/mol; (18)Boiling Point: 582 °C at 760 mmHg; (19)Vapour Pressure: 5.69E-16 mmHg at 25°C.
Uses of Prednisone 21-acetate: it can be used to produce 21-acetoxy-1α,2α-epoxy-17α-hydroxy-4-pregnene-3,11,20-trione at temperature of 20 °C. This reaction will need reagent dimethyldioxirane and solvent acetone with reaction time of 20 hours. The yield is about 80%.

You can still convert the following datas into molecular structure:
(1)SMILES: O=C(OCC(=O)[C@@]3(O)CC[C@H]2[C@@H]4CC\C1=C\C(=O)\C=C/[C@]1(C)[C@H]4C(=O)C[C@@]23C)C
(2)InChI: InChI=1/C23H28O6/c1-13(24)29-12-19(27)23(28)9-7-17-16-5-4-14-10-15(25)6-8-21(14,2)20(16)18(26)11-22(17,23)3/h6,8,10,16-17,20,28H,4-5,7,9,11-12H2,1-3H3/t16-,17-,20+,21-,22-,23-/m0/s1
(3)InChIKey: MOVRKLZUVNCBIP-RFZYENFJBM