Products Categories
| CAS No.: | 1633-05-2 |
|---|---|
| Name: | Strontium carbonate |
| Article Data: | 213 |
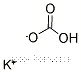
| Molecular Structure: | |
|
|
|
| Formula: | SrCO3 |
| Molecular Weight: | 147.629 |
| Synonyms: | Strontium carbonate (SrCO3);Strontianite;NSC 112224;HSDB 5845;Carbonic acid strontium salt (1:1);CI 77837;CCRIS 3203;C.I. 77837; |
| EINECS: | 216-643-7 |
| Density: | 3.7 g/mL at 25 °C(lit.) |
| Melting Point: | 1497 °C |
| Boiling Point: | 333.6 °C at 760 mmHg |
| Flash Point: | 169.8 °C |
| Appearance: | Milky white free flowing powder |
| PSA: | 63.19000 |
| LogP: | -2.44700 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 211914-50-0N-[[2-[[[4-(Aminoiminomethyl)phenyl]amino]methyl]-1-methyl-1H-benzimidazol-5-yl]carbonyl]-N-(2-pyridinyl)-beta-alanine ethyl ester hydrochloride
- 95-48-7Phenol,2-methyl-
- 87-33-2Isosorbide dinitrate
- 611-06-32,4-Dichloronitrobenzene
- 9000-70-8Gelatin
- 108-11-22-Pentanol, 4-methyl-
- 80-56-8Bicyclo[3.1.1]hept-2-ene,2,6,6-trimethyl-
- 15833-61-13-Furanmethanol,tetrahydro-
- 73334-07-3Iopromide
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
Analytical Methods
Strontium carbonate (CAS NO.1633-05-2) is prepared synthetically in one of two manners. First of which is from naturally occurring celestine by the reaction in solution with a soluble carbonate salt (usually sodium or ammonium carbonates).
For example if sodium carbonate was used in solution with strontium nitrate:
Sr(NO3)2 (aq) + Na2CO3 (aq) → SrCO3 (s) + 2 NaNO3 (aq).
Specification
Strontium carbonate(CAS NO.1633-05-2) is the carbonate salt of strontium that has the appearance of a white or grey powder. It occurs in nature as the mineral strontianite.Being a carbonate, it is a weak base and therefore is reactive with acids. It is otherwise stable and safe to work with. It is practically insoluble in water (1 part in 100,000). The solubility is increased significantly if the water is saturated with carbon dioxide, to 1 part in 1,000 ,also it is soluble in dilute acids.
Physical properties about Strontium carbonate are: (1)ACD/LogP: -0.809; (2)ACD/LogD (pH 5.5): -1.98; (3)ACD/LogD (pH 7.4): -3.77; (4)ACD/BCF (pH 5.5): 1.00; (5)ACD/BCF (pH 7.4): 1.00; (6)ACD/KOC (pH 5.5): 1.00; (7)ACD/KOC (pH 7.4):1.00; (8)#H bond acceptors: 3; (9)#H bond donors: 2; (10)Flash Point: 169.8 °C; (11)Enthalpy of Vaporization: 63.37 kJ/mol; (12)Boiling Point: 333.6 °C at 760 mmHg; (13)Vapour Pressure: 2.58E-05 mmHg at 25°C
Preparation of Strontium carbonate: Strontium carbonate is prepared synthetically in one of two manners. First of which is from naturally occurring celestine also known as strontium sulfate (SrSO4) or by using soluble strontium salts by the reaction in solution with a soluble carbonate salt (usually sodium or ammonium carbonates). For example if sodium carbonate was used in solution with strontium nitrate:
Sr(NO3)2 (aq) + Na2CO3 (aq) → SrCO3 (s) + 2 NaNO3 (aq).
Uses of Strontium carbonate: The most common use is as an inexpensive colorant in fireworks. Strontium and its salts emit a brilliant red color in flame.Strontium carbonate is used for electronic applications. It is used for manufacturing CTV to absorb electrons resulting from the cathode. It is used in the preparation of iridescent glass, luminous paints, strontium oxide or strontium salts and in refining sugar and certain drugs.
You can still convert the following datas into molecular structure:
(1)InChI=1S/CH2O3.Sr/c2-1(3)4;/h(H2,2,3,4);/q;+2/p-2;
(2)InChIKey=LEDMRZGFZIAGGB-UHFFFAOYSA-L;
(3)SmilesC(=O)([O-])[O-].[Sr+2];