Products Categories
| CAS No.: | 22862-76-6 |
|---|---|
| Name: | ANISOMYCIN |
| Article Data: | 8 |
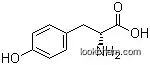
| Molecular Structure: | |
|
|
|
| Formula: | C14H19NO4 |
| Molecular Weight: | 265.309 |
| Synonyms: | 3,4-Pyrrolidinediol,2-(p-methoxybenzyl)-, 3-acetate, (2R,3S,4S)- (8CI);3,4-Pyrrolidinediol,2-[(4-methoxyphenyl)methyl]-, 3-acetate, [2R-(2a,3a,4b)]-;Anisomycin (6CI,7CI);(-)-Anisomycin;(2R,3S,4S)-2-(p-Methoxybenzyl)-3,4-pyrrolidinediol 3-acetate;(2R,3S,4S)-2-(p-Methoxyphenylmethyl)-3-acetoxy-4-hydroxypyrrolidine;Anisomycin, (-)-;Flagecidin;NSC 76712; |
| EINECS: | 245-269-7 |
| Density: | 1.21 g/cm3 |
| Melting Point: | 140-141 °C |
| Boiling Point: | 398.7 °C at 760 mmHg |
| Flash Point: | 194.9 °C |
| Solubility: | Soluble in water at 2 mg/ml, in DMSO at 20mg/ml, and in methanol at 20mg/ml |
| Appearance: | White crystalline solid |
| Hazard Symbols: |
 T, T,  Xn Xn
|
| Risk Codes: | 25-36/37/38-20/21/22 |
| Safety: | 45-36-26 |
| Transport Information: | UN 3462 6.1/PG 3 |
| PSA: | 67.79000 |
| LogP: | 0.83090 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 240444-70-62,3,5,6-Tetrafluor-4-(methoxymethyl)benzyl-2,2-dimethyl-3-(prop-1-en-1-yl)cyclopropancarboxylat
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 27958-08-3
(2R,3S,4S)-3-acetoxy-N-(benzyloxycarbonyl)-4-hydroxy-2-(4-methoxybenzyl)pyrrolidine

- 22862-76-6
(-)-anisomycin

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethanol under 760 Torr; for 0.25h; | 95% |
- 180081-83-8
(2R,3S,4S)-3-Acetoxy-1-(tert-butoxycarbonyl)-4-(tert-butyldimethylsilyloxy)-2-(4-methoxybenzyl)pyrrolidine

- 22862-76-6
(-)-anisomycin

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol at 20℃; | 88% |
- 1262850-97-4
C19H27NO6

- 22862-76-6
(-)-anisomycin

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol | 78% |
- 933992-79-1
(2R,3S,4S)-3-acetoxy-1-benzyl-4-hydroxy-2-(4-methoxybenzyl)-pyrrolidine

- 22862-76-6
(-)-anisomycin

| Conditions | Yield |
|---|---|
| With hydrogenchloride; hydrogen; palladium on activated charcoal In methanol; water for 3h; Yield given; |
- 87796-48-3
(2R,3S,4S)-3-acetoxy-4-allyloxy-1-benzyl-2-(p-methoxybenzyl)pyrrolidine

- 22862-76-6
(-)-anisomycin

| Conditions | Yield |
|---|---|
| With hydrogenchloride; hydrogen; palladium on activated charcoal 1.) MeOH, reflux, 2.) MeOH; Yield given. Multistep reaction; | |
| Multi-step reaction with 2 steps 1: HCl, water / palladium-charcoal 10percent / methanol / 48 h / Heating 2: HCl, H2 / palladium-charcoal 10percent / methanol; H2O / 3 h View Scheme |
- 173266-54-1
Acetic acid (1S,2R)-2-azido-1-((R)-2-bromo-1-hydroxy-ethyl)-3-(4-methoxy-phenyl)-propyl ester

- 22862-76-6
(-)-anisomycin

| Conditions | Yield |
|---|---|
| With sodium acetate In methanol for 10h; Heating; Yield given; |

- 22862-76-6
(-)-anisomycin

| Conditions | Yield |
|---|---|
| With hydrogen; palladium dihydroxide In methanol; acetic acid at 20℃; under 3878.71 Torr; for 24h; | 76 mg |
- 951385-80-1
(4R,5S,6S)-5-(tert-butyldimethylsilyloxy)-4-(4-methoxybenzyl)-2-phenyl-6-vinyl-5,6-dihydro-4H-1,3-oxazine

- 22862-76-6
(-)-anisomycin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 89 percent / Bu4NF / tetrahydrofuran / 1 h / 20 °C 2: 98 percent / pyridine / CH2Cl2 3: O3; O2 / methanol / -78 °C 4: 76 mg / H2 / Pd(OH)2 / methanol; acetic acid / 24 h / 20 °C / 3878.71 Torr View Scheme |
- 951385-97-0
C20H21NO3

- 22862-76-6
(-)-anisomycin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 98 percent / pyridine / CH2Cl2 2: O3; O2 / methanol / -78 °C 3: 76 mg / H2 / Pd(OH)2 / methanol; acetic acid / 24 h / 20 °C / 3878.71 Torr View Scheme |
- 951385-93-6
(4R,5S,6S)-4-(4-methoxybenzyl)-2-phenyl-6-vinyl-5,6-dihydro-4H-1,3-oxazin-5-yl acetate

- 22862-76-6
(-)-anisomycin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: O3; O2 / methanol / -78 °C 2: 76 mg / H2 / Pd(OH)2 / methanol; acetic acid / 24 h / 20 °C / 3878.71 Torr View Scheme |
Chemistry
Molecular Formula: C14H19NO4
Molar mass: 265.305 g/mol
EINECS: 245-269-7
Form: Solid
Color: White
Density: 1.21 g/cm3
Flash Point: 194.9 °C
Melting point: 140-141 °C
Storage temp: 2-8 °C
Appearance: Crystalline
Index of Refraction: 1.557
Boiling Point: 398.7 °C at 760 mmHg
Vapour Pressure: 4.5E-07 mmHg at 25 °C
Solubility: Methanol: 20 mg/mL, clear, colorless to faintly yellow
Stable: Stable. Incompatible with strong oxidizing agents.
Product categories of Anisomycin (CAS NO.22862-76-6): Antibiotics;Protein Kinase
Structure of Anisomycin (CAS NO.22862-76-6):

Uses
Anisomycin (CAS NO.22862-76-6) has wide usage as a protein biosynthesis inhibitor. It is used as a component of Martin Lewis Agar, an in- vitro diagnostic product which is used extensively in the United States for the selective isolation of Neisseria gonorrhoeae and Neisseria meningitidis. Mawji et al. showed that anisomycin can sensitize metastatic epithelial cells to anoikis and reduce circulating tumor cell implantation in vivo by using a novel high-throughput screening assay. Anisomycin achieved this anti-metastatic activity in part by decreasing the abundance of the death receptor inhibiting protein FLIP.
Production
Although pyrrolidine-based structure of Anisomycin (CAS NO.22862-76-6) suggests that it is derived from proline, the results from the experiments indicated that tyrosine, glycine, methionine, and acetate are the primary precursors for the biosynthesis of anisomycin. The methord is:
.png)
Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| cat | LD | oral | > 200mg/kg (200mg/kg) | GASTROINTESTINAL: NAUSEA OR VOMITING | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. |
| dog | LD | intramuscular | > 100mg/kg (100mg/kg) | GASTROINTESTINAL: NAUSEA OR VOMITING | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. |
| dog | LD | intravenous | > 100mg/kg (100mg/kg) | GASTROINTESTINAL: NAUSEA OR VOMITING | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. |
| dog | LD | oral | > 300mg/kg (300mg/kg) | GASTROINTESTINAL: NAUSEA OR VOMITING | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. |
| guinea pig | LDLo | oral | 300mg/kg (300mg/kg) | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. | |
| monkey | LD | intramuscular | > 50mg/kg (50mg/kg) | GASTROINTESTINAL: NAUSEA OR VOMITING | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. |
| monkey | LD | oral | > 300mg/kg (300mg/kg) | GASTROINTESTINAL: NAUSEA OR VOMITING | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. |
| monkey | LDLo | intravenous | 200mg/kg (200mg/kg) | GASTROINTESTINAL: NAUSEA OR VOMITING | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. |
| mouse | LD50 | intraperitoneal | 400mg/kg (400mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD LUNGS, THORAX, OR RESPIRATION: RESPIRATORY DEPRESSION | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. |
| mouse | LD50 | intravenous | 10mg/kg (10mg/kg) | "CRC Handbook of Antibiotic Compounds," Vols.1- , Berdy, J., Boca Raton, FL, CRC Press, 1980Vol. 5, Pg. 77, 1981. | |
| mouse | LD50 | oral | 148mg/kg (148mg/kg) | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. | |
| mouse | LD50 | subcutaneous | 600mg/kg (600mg/kg) | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. | |
| rabbit | LD | oral | > 200mg/kg (200mg/kg) | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. | |
| rat | LD50 | intraperitoneal | 345mg/kg (345mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD LUNGS, THORAX, OR RESPIRATION: RESPIRATORY DEPRESSION | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. |
| rat | LD50 | intravenous | 167mg/kg (167mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD LUNGS, THORAX, OR RESPIRATION: RESPIRATORY DEPRESSION | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. |
| rat | LD50 | oral | 72mg/kg (72mg/kg) | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. | |
| rat | LD50 | subcutaneous | 230mg/kg (230mg/kg) | Antibiotics and Chemotherapy Vol. 5, Pg. 490, 1955. |
Safety Profile
Poison by ingestion, intraperitoneal, subcutaneous, and intravenous routes. When heated to decomposition it emits toxic fumes of NOx.
Hazard Codes:  T
T Xn
Xn
Risk Statements: 25-36/37/38-20/21/22
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed.
R25 :Toxic if swallowed.
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 45-36-26
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
Specification
Anisomycin ,its cas register number is 22862-76-6. It also can be called Flagecidin ; 4-Hydroxy-2-(4-methoxybenzyl)-3-pyrrolidinylacetat ; Acétate de 4-hydroxy-2-(4-méthoxybenzyl)-3-pyrrolidinyle and 3,4-pyrrolidinediol, 2-[(4-methoxyphenyl)methyl]-, 3-acetate . It is inactive against bacteria, and can activate stress-activated protein kinases, MAP kinase and other signal transduction pathways. Partial inhibition of DNA synthesis occurs at anisomycin concentrations that effect 95% inhibition of protein synthesis.