Products Categories
| CAS No.: | 32383-76-9 |
|---|---|
| Name: | Medicarpin |
| Article Data: | 5 |
| Molecular Structure: | |
|
|
|
| Formula: | C16H14O4 |
| Molecular Weight: | 270.285 |
| Synonyms: | (-)-3-Hydroxy-9-methoxypterocarpan;(-) Medicarpin; |
| Density: | 1.319 g/cm3 |
| Melting Point: | 127.5-128.5° |
| Boiling Point: | 418.8 °C at 760 mmHg |
| Flash Point: | 207.1 °C |
| PSA: | 47.92000 |
| LogP: | 3.01050 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 485-72-3
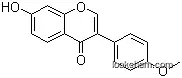
7-Hydroxy-3-(4-methoxy-phenyl)-chromen-4-on

- 32383-76-9
medicarpin
- 770722-02-6
2,7-dihydroxy-4'-methoxyisoflavanone

- 32383-76-9
medicarpin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 2-hydroxyisoflavanone dehydratase View Scheme |
- 131887-80-4
(2R,3S)-2,7,4'-trihydroxyisoflavanone

- 32383-76-9
medicarpin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 2-methoxyethanol / 2,7,4'-trihydroxyisoflavanone 4'-O-methyltransferase 2: 2-hydroxyisoflavanone dehydratase View Scheme |
- 1891-01-6
3-(2,4-dimethoxyphenyl)-7-hydroxy-4H-chromen-4-one

A
- 32383-76-9
medicarpin

B
- 33983-39-0
medicarpin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: aluminum (III) chloride / acetonitrile / 0 °C / Reflux 1.2: Cooling with ice 2.1: sodium tetrahydroborate / ethanol / 0 - 20 °C 3.1: Lux cellulose-1 PHENOMENIX column / acetonitrile; water / Resolution of racemate View Scheme |
- 1890-99-9
7-hydroxy-3-(2-hydroxy-4-methoxyphenyl)-4H-chromen-4-one

A
- 32383-76-9
medicarpin

B
- 33983-39-0
medicarpin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium tetrahydroborate / ethanol / 0 - 20 °C 2: Lux cellulose-1 PHENOMENIX column / acetonitrile; water / Resolution of racemate View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: boron trifluoride diethyl etherate / 2 h / 90 - 100 °C 1.2: 50 - 90 °C 2.1: aluminum (III) chloride / acetonitrile / 0 °C / Reflux 2.2: Cooling with ice 3.1: sodium tetrahydroborate / ethanol / 0 - 20 °C 4.1: Lux cellulose-1 PHENOMENIX column / acetonitrile; water / Resolution of racemate View Scheme |
- 32383-76-9, 33983-39-0, 33983-40-3, 57458-34-1
9-methoxy-6a,11a-dihydro-6H-benzo[4,5]furo[3,2-c]chromen-3-ol

A
- 32383-76-9
medicarpin

B
- 33983-39-0
medicarpin

| Conditions | Yield |
|---|---|
| With Lux cellulose-1 PHENOMENIX column In water; acetonitrile Resolution of racemate; | A n/a B n/a |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: boron trifluoride diethyl etherate / 2 h / 90 - 100 °C 1.2: 50 - 90 °C 2.1: aluminum (III) chloride / acetonitrile / 0 °C / Reflux 2.2: Cooling with ice 3.1: sodium tetrahydroborate / ethanol / 0 - 20 °C 4.1: Lux cellulose-1 PHENOMENIX column / acetonitrile; water / Resolution of racemate View Scheme |

- 32383-76-9
medicarpin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: sodium hydride / N,N-dimethyl-formamide / 5 h / -78 - 20 °C / Inert atmosphere 2: triethylsilane; boron trifluoride diethyl etherate / dichloromethane / 2.17 h / -78 °C 3: lead(II,IV) oxide / benzene / 3 h / 85 °C 4: palladium 10% on activated carbon; hydrogen / ethanol 5: potassium carbonate / ethyl acetate / 5 h / 20 °C View Scheme |

- 32383-76-9
medicarpin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: triethylsilane; boron trifluoride diethyl etherate / dichloromethane / 2.17 h / -78 °C 2: lead(II,IV) oxide / benzene / 3 h / 85 °C 3: palladium 10% on activated carbon; hydrogen / ethanol 4: potassium carbonate / ethyl acetate / 5 h / 20 °C View Scheme |
Specification
The Medicarpin, with the CAS registry number 32383-76-9, is also known as (-)-3-Hydroxy-9-methoxypterocarpan. This chemical's molecular formula is C16H14O4 and molecular weight is 270.28. What's more, its IUPAC name is (6aR, 11aR)-9-Methoxy-6a, 11a-dihydro-6H-[1]benzofuro[3, 2-c]chromen-3-ol. In addition, Medicarpin is a pterocarpan, a derivative of isoflavonoids, found in Medicago truncatula.
Physical properties about Medicarpin are: (1)ACD/LogP: 2.72; (2)# of Rule of 5 Violations: 0; (3)#H bond acceptors: 4; (4)#H bond donors: 1; (5)#Freely Rotating Bonds: 2; (6)Polar Surface Area: 36.92 Å2; (7)Index of Refraction: 1.629; (8)Molar Refractivity: 72.84 cm3; (9)Molar Volume: 204.8 cm3; (10)Polarizability: 28.87×10-24 cm3; (11)Surface Tension: 54.4 dyne/cm; (12)Density: 1.319 g/cm3; (13)Flash Point: 207.1 °C; (14)Enthalpy of Vaporization: 69.85 kJ/mol; (15)Boiling Point: 418.8 °C at 760 mmHg; (16)Vapour Pressure: 1.31E-07 mmHg at 25 °C.
You can still convert the following datas into molecular structure:
(1) SMILES: O3c1c(ccc(O)c1)[C@@H]4Oc2cc(OC)ccc2[C@@H]4C3
(2) InChI: InChI=1/C16H14O4/c1-18-10-3-5-11-13-8-19-14-6-9(17)2-4-12(14)16(13)20-15(11)7-10/h2-7,13,16-17H,8H2,1H3/t13-,16-/m0/s1
(3) InChIKey: NSRJSISNDPOJOP-BBRMVZONBK