Products Categories
| CAS No.: | 502-42-1 |
|---|---|
| Name: | Cycloheptanone |
| Article Data: | 367 |
| Molecular Structure: | |
|
|
|
| Formula: | C7H12O |
| Molecular Weight: | 112.172 |
| Synonyms: | Ketocycloheptane;Ketoheptamethylene;NSC 9471;Suberon;Suberone; |
| EINECS: | 207-937-6 |
| Density: | 0.929 g/cm3 |
| Melting Point: | -21 °C |
| Boiling Point: | 180 °C at 760 mmHg |
| Flash Point: | 55.6 °C |
| Solubility: | Insoluble in water |
| Appearance: | clear colorless to yellow liquid |
| Hazard Symbols: |
 Xi Xi
|
| Risk Codes: | 10-36/37/38 |
| Safety: | 23-24/25-36-26 |
| Transport Information: | UN 1987 3/PG 3 |
| PSA: | 17.07000 |
| LogP: | 1.90970 |
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 717878-06-31-(4-fluorophenyl)-4-nitro-1H-imidazole

| Conditions | Yield |
|---|---|
| With ruthenium trichloride; iodobenzene; potassium peroxomonosulfate In water; acetonitrile at 20℃; for 0.8h; | 100% |
| With ruthenium trichloride; iodobenzene; potassium peroxymonosulfate In water; acetonitrile at 20℃; for 0.8h; Inert atmosphere; | 100% |
| With 5 wt% Pd nanoparticles loaded on phosphate anion exchanged [Mg6Al2(OH)16]CO3*xH2O; air at 50℃; under 760.051 Torr; for 6h; Reagent/catalyst; Irradiation; | 100% |
- 36237-80-6
cycloheptanone semicarbazone

- 502-42-1
cycloheptanone

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In tetrahydrofuran; methanol; water for 3h; Heating; | 100% |
- 77378-96-2
1-cycloheptylidenehydrazine

- 502-42-1
cycloheptanone

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In tetrahydrofuran; methanol; water for 2h; Heating; | 100% |
| With HOF* CH3CN In dichloromethane at 0℃; for 0.0166667h; |

| Conditions | Yield |
|---|---|
| With hydrogen; palladium(II) complex of ferrocenylamine sulfide (3) In acetone under 4137.2 Torr; for 8h; | 95% |
| With hydrogen In ethanol at 20℃; under 760.051 Torr; for 1h; chemoselective reaction; | 94% |
| With hydrogen; palladium |

| Conditions | Yield |
|---|---|
| With Oxone; water; potassium hydrogencarbonate In acetone for 0.25h; Heating; | 95% |
| With Cu(NO3)2-SiO2 for 0.166667h; oxime cleavage; microwave irradiation; | 95% |
| With γ-picolinium chlorochromate; silica gel In dichloromethane at 20℃; for 4h; | 93% |
- 25632-02-4
1,1-dimethoxycycloheptane

- 502-42-1
cycloheptanone

| Conditions | Yield |
|---|---|
| With Methyltrichlorosilane; sodium iodide In acetonitrile for 0.5h; Ambient temperature; | 95% |
- 1056246-49-1
2-bromocycloheptanone

- 502-42-1
cycloheptanone

| Conditions | Yield |
|---|---|
| With hydrogen iodide for 0.0833333h; Ambient temperature; | 93% |
| With chloro-trimethyl-silane; sodium iodide In acetonitrile for 8h; Ambient temperature; | 88% |
- 64-18-6
formic acid

- 3349-69-7
1-cycloheptylidene-2-phenylhydrazine

A
- 622-84-4
2-formyl-1-phenylhydrazine

B
- 502-42-1
cycloheptanone

| Conditions | Yield |
|---|---|
| In ethanol for 5h; Heating; | A 93% B 87% |

- 3349-69-7
1-cycloheptylidene-2-phenylhydrazine

A
- 622-84-4
2-formyl-1-phenylhydrazine

B
- 502-42-1
cycloheptanone

| Conditions | Yield |
|---|---|
| With formic acid In ethanol for 5h; Heating; | A 93% B 87% |
- 72170-88-8
Cycloheptanone SAMP-hydrazone

- 502-42-1
cycloheptanone

| Conditions | Yield |
|---|---|
| With selenium(IV) oxide; dihydrogen peroxide In methanol; water at 20℃; pH=7; aq. phosphate buffer; | 93% |
Chemistry
Molecular Formula: C7H12O
Molecular Weight: 112.17
EINECS: 207-937-6
Melting point: -21 °C
Boiling Point: 180 °C at 760 mmHg
Flash Point: 55.6 °C
Index of Refraction: 1.449
Molar Refractivity: 32.41 cm3
Molar Volume: 120.6 cm3
Surface Tension: 31.5 dyne/cm
Density: 0.929 g/cm3
Enthalpy of Vaporization: 41.63 kJ/mol
Vapour Pressure: 0.915 mmHg at 25°C
Storage temp.: Flammables area
Water Solubility: Insoluble in water
Appearance: Clear colorless to yellow liquid
Structure of Suberone (CAS NO.502-42-1):
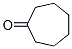
IUPAC Name: Cycloheptanone
Canonical SMILES: C1CCCC(=O)CC1
InChI: InChI=1S/C7H12O/c8-7-5-3-1-2-4-6-7/h1-6H2
InChIKey: CGZZMOTZOONQIA-UHFFFAOYSA-N
Product Category of Suberone (CAS NO.502-42-1): Pharmaceutical Intermediates;Carbonyl Compounds;Ketones
Uses
Suberone (CAS NO.502-42-1) is used in organic synthesis.
Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LDLo | intraperitoneal | 750mg/kg (750mg/kg) | BEHAVIORAL: EXCITEMENT BEHAVIORAL: COMA CARDIAC: ARRHYTHMIAS (INCLUDING CHANGES IN CONDUCTION) | Comptes Rendus Hebdomadaires des Seances, Academie des Sciences. Vol. 254, Pg. 2245, 1962. |
| mouse | LDLo | subcutaneous | 930mg/kg (930mg/kg) | AUTONOMIC NERVOUS SYSTEM: OTHER (DIRECT) PARASYMPATHOMIMETIC BEHAVIORAL: COMA LUNGS, THORAX, OR RESPIRATION: RESPIRATORY DEPRESSION | Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 50, Pg. 199, 1903. |
Safety Profile
Moderately toxic by subcutaneous and intraperitoneal routes. Can cause central nervous system depression. When heated to decomposition it emits acrid smoke and irritating fumes.
The Hazard Codes:  Xi
Xi
Hazard Note: Irritant
HazardClass: 3
The Risk Statements:
10: Flammable
36/37/38: Irritating to eyes, respiratory system and skin
The Safety Statements:
23: Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer)
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
24/25: Avoid contact with skin and eyes
RIDADR: UN 1987 3/PG 3
WGK Germany: 3
RTECS: GU3325000
PackingGroup: III
Specification
Suberone , its cas register number is 502-42-1. It also can be called Cycloheptanone ; Ketoheptamethylene ; and Suberon . It is hazardous, so the first aid measures and others should be known. Such as: When on the skin: first, should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly, get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While, it's inhaled: Remove from exposure and move to fresh air immediately. Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product: Wash mouth out with water, and get medical aid immediately. Notes to physician: Treat supportively and symptomatically.
In addition, Suberone (CAS NO.502-42-1) is not compatible with strong oxidizing agents, reducing agents, strong bases, and you must not take it with incompatible materials, ignition sources. And also prevent it to broken down into hazardous decomposition products: Carbon monoxide, carbon dioxide.

