Products Categories
| CAS No.: | 521-17-5 |
|---|---|
| Name: | Androstenediol |
| Article Data: | 91 |
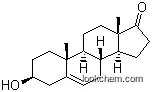
| Molecular Structure: | |
|
|
|
| Formula: | C19H30O2 |
| Molecular Weight: | 290.446 |
| Synonyms: | Androst-5-ene-3b,17b-diol (7CI,8CI);3b,17b-Androst-5-enediol;3b,17b-Dihydroxyandrost-5-ene;Androst-5-enediol;Hermaphrodiol;NSC 12163;D5-Androstene-3b,17b-diol;D5-Androstenediol; |
| EINECS: | 208-306-8 |
| Density: | 1.12 g/cm3 |
| Melting Point: | 178-182 °C |
| Boiling Point: | 428.4 °C at 760 mmHg |
| Flash Point: | 194.5 °C |
| Appearance: | white crystalline powder |
| Hazard Symbols: | F,T |
| Risk Codes: | 60-61-11-19-38 |
| Safety: | 24/25 |
| PSA: | 40.46000 |
| LogP: | 3.67100 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 717878-06-31-(4-fluorophenyl)-4-nitro-1H-imidazole
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 548773-13-32-amino-4,6-dimethylpyrimidine-5-carboxylic acid
- 1461-22-9Stannane,tributylchloro-
- 540-51-2Ethanol, 2-bromo-
- 116649-85-5Ramatroban
- 57644-54-91,2,3-Propanetricarboxylicacid, 2-hydroxy-, bismuth(3+) potassium salt (2:1:3)
- 136-52-7Hexanoic acid,2-ethyl-, cobalt(2+) salt (2:1)
- 69604-00-82-Benzofurancarboxylicacid, 5-nitro-, ethyl ester
- 39711-79-0Cyclohexanecarboxamide,N-ethyl-5-methyl-2-(1-methylethyl)-
- 12007-60-2Lithium tetraborate
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
Specification
The IUPAC name of Androstenediol is (3S,8R,9S,10R,13S,14S,17S)-10,13-dimethyl-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthrene-3,17-diol. With the CAS registry number 521-17-5, it is also named as (3-beta,17-beta)-Androst-5-ene-3,17-diol. The classification codes are Anabolic Agents; Drug / Therapeutic Agent; Hormones, Hormone Substitutes, and Hormone Antagonists; Reproductive Effect. It is white crystalline powder which is one of two androstenediols. Additionally, this chemical must be sealed in the container and the container should be placed in the cool and dry aera.
The other characteristics of this product can be summarized as: (1)ACD/LogP: 3.96; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 3.96; (4)ACD/LogD (pH 7.4): 3.96; (5)ACD/BCF (pH 5.5): 603.66; (6)ACD/BCF (pH 7.4): 603.66; (7)ACD/KOC (pH 5.5): 3404.83; (8)ACD/KOC (pH 7.4): 3404.83; (9)#H bond acceptors: 2; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 2; (12)Index of Refraction: 1.568; (13)Molar Refractivity: 84.5 cm3; (14)Molar Volume: 258 cm3; (15)Polarizability: 33.5×10-24 cm3; (16)Surface Tension: 46.4 dyne/cm; (17)Enthalpy of Vaporization: 78.92 kJ/mol; (18)Vapour Pressure: 3.91E-09 mmHg at 25°C; (19)Exact Mass: 290.22458; (20)MonoIsotopic Mass: 290.22458; (21)Topological Polar Surface Area: 40.5; (22)Heavy Atom Count: 21.
Preparation of Androstenediol: It is derived from dehydroepiandrosterone by the reduction of the 17-keto group (17-hydroxysteroid dehydrogenases), is converted to testosterone by the oxidation of the 3-beta hydroxyl group to a 3-keto group (3-hydroxysteroid dehydrogenases).
Uses of Androstenediol: It is used as intermediate of steroid hormone and acyeterion. Its potential use as a radiation countermeasure was introduced by the Armed Forces Radiobiology Research Institute (AFRRI) and subsequently studied by AFRRI and Hollis-Eden Pharmaceuticals under the tradename Neumune for the treatment of acute radiation syndrome.
People can use the following data to convert to the molecule structure.
1. SMILES:O[C@@H]4C/C3=C/C[C@@H]1[C@H](CC[C@@]2([C@@H](O)CC[C@@H]12)C)[C@@]3(C)CC4
2. InChI:InChI=1/C19H30O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h3,13-17,20-21H,4-11H2,1-2H3/t13-,14-,15-,16-,17-,18-,19-/m0/s1.