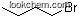Products Categories
| CAS No.: | 544-16-1 |
|---|---|
| Name: | 1-Butyl nitrite |
| Article Data: | 31 |
| Molecular Structure: | |
|
|
|
| Formula: | C4H9NO2 |
| Molecular Weight: | 103.121 |
| Synonyms: | UNII-44P8QG0F3T;Butyl nitrite;BRN 1701036;NSC 8426;NCI-C56553;CCRIS 838; |
| EINECS: | 208-862-1 |
| Density: | 0.99 g/cm3 |
| Boiling Point: | 78.8 °C at 760 mmHg |
| Flash Point: | -13.333 °C |
| Solubility: | Slightly soluble in water |
| Appearance: | Clear yellow liquid |
| Hazard Symbols: |
 F, F, T T
|
| Risk Codes: | 11-23/25 |
| Safety: | 16-24-45 |
| Transport Information: | UN 2351 3/PG 2 |
| PSA: | 38.66000 |
| LogP: | 1.48450 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 717878-06-31-(4-fluorophenyl)-4-nitro-1H-imidazole
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 548773-13-32-amino-4,6-dimethylpyrimidine-5-carboxylic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid; sodium nitrite In water at 0℃; for 1h; | 80% |
| With tert.-butylnitrite In chloroform at 26℃; Equilibrium constant; | |
| With nitrosylchloride In tetrachloromethane; acetic acid at 5℃; Thermodynamic data; Equilibrium constant; Rate constant; Ea, ΔH, ΔG, ΔS; various temperatures and solvent composition; solvent-jump relaxation; mechanism; |

| Conditions | Yield |
|---|---|
| With tetraphenylphosphonium nitrite (ionic form) Yield given. Yields of byproduct given; | |
| With silver(I) nitrite; diethyl ether | |
| With silver(I) nitrite; Petroleum ether | |
| With tetraphenylphosphonium nitrite (ionic form) Yield given. Yields of byproduct given; |

| Conditions | Yield |
|---|---|
| With tetraphenylphosphonium nitrite (ionic form) Yield given. Yields of byproduct given; | |
| With silver(I) nitrite; Petroleum ether | |
| With silver(I) nitrite; diethyl ether | |
| With tetraphenylphosphonium nitrite (ionic form) Yield given. Yields of byproduct given; |

| Conditions | Yield |
|---|---|
| With dichloromethane; dinitrogen tetraoxide; butan-1-ol |

| Conditions | Yield |
|---|---|
| With H2(15)NO at 175℃; Product distribution; ratios of disproportion to combination reactions; labeled by 15N; |

| Conditions | Yield |
|---|---|
| With Cu(NO3)2-SiO2 In tetrachloromethane for 1h; Heating; Yield given. Yields of byproduct given; |
- 106-97-8
n-butane

A
- 544-16-1
n-Butyl nitrite

B
- 928-45-0
butyl nitrate

C
- 924-43-6
sec-butyl nitrite

D
- 600-24-8, 116783-22-3
2-nitrobutane

E
- 924-52-7
2-butyl nitrate

F
- 627-05-4
1-nitrobutane

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; Nitrogen dioxide at 24.9℃; under 300.02 Torr; Rate constant; Product distribution; Mechanism; boric-acid-coated surface; various pressure and carrier-gas composition; | A 6 % Chromat. B 0.3 % Chromat. C 31 % Chromat. D 51 % Chromat. E 3 % Chromat. F 7 % Chromat. |



Consensus Reports
Specification
The Butyl nitrite with CAS registry number of 544-16-1 is also known as Nitrous acid, butyl ester. The IUPAC name is Butyl nitrite. It belongs to product categories of Organics; Carboxylic; Nitrogen Compounds; Organic Building Blocks; Organic Nitrates/Nitrites. Its EINECS registry number is 208-862-1. In addition, the formula is C4H9NO2 and the molecular weight is 103.12. This chemical is a clear yellow liquid and should be stored at the temperature of 0-6 °C.
Physical properties about Butyl nitrite are: (1)ACD/LogP: 2.11; (2)ACD/LogD (pH 5.5): 2.1; (3)ACD/LogD (pH 7.4): 2.1; (4)ACD/BCF (pH 5.5): 23.41; (5)ACD/BCF (pH 7.4): 23.41; (6)ACD/KOC (pH 5.5): 332.58; (7)ACD/KOC (pH 7.4): 332.58; (8)#H bond acceptors: 3; (9)#Freely Rotating Bonds: 4; (10)Index of Refraction: 1.415; (11)Molar Refractivity: 26.08 cm3; (12)Molar Volume: 104 cm3; (13)Surface Tension: 31.1 dyne/cm; (14)Density: 0.99 g/cm3; (15)Enthalpy of Vaporization: 30.64 kJ/mol; (16)Boiling Point: 78.8 °C at 760 mmHg; (17)Vapour Pressure: 101 mmHg at 25 °C.
Preparation of Butyl nitrite: it is prepared by reaction of butan-1-ol. The reaction needs reagents NaNO2, H2SO4 and solvent H2O at the temperature of 0 °C for 1 hour. The yield is about 80%.
![]()
Uses of Butyl nitrite: it can be used as solvent, analytical reagent and organic synthesis intermediate. Besides, it is used to produce N-nitroso-N-(n-butoxymethyl)-2-chlorophenylamine by reaction with 1,3,5-tris-(2-chloro-phenyl)-hexahydro-[1,3,5]triazine. The reaction occurs with solvent CH2Cl2 and other condition of heating for 2 hours. The yield is about 70%.
![Butyl nitrite is used to produce N-nitroso-N-(n-butoxymethyl)-2-chlorophenylamine by reaction with 1,3,5-tris-(2-chloro-phenyl)-hexahydro-[1,3,5]triazine.](/UserFilesUpload/Uses of Butyl nitrite.png)
When you are using this chemical, please be cautious about it. As a chemical, it is highly flammable that you keep away from sources of ignition. This chemical is also toxic by inhalation and if swallowed. During using it, avoid contact with skin. In case of accident or if you feel unwell seek medical advice immediately.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: CCCCON=O
2. InChI: InChI=1S/C4H9NO2/c1-2-3-4-7-5-6/h2-4H2,1H3
3. InChIKey: JQJPBYFTQAANLE-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| man | TDLo | oral | 153mg/kg (153mg/kg) | BLOOD: METHEMOGLOBINEMIA-CARBOXYHEMOGLOBIN | Annals of Internal Medicine. Vol. 92, Pg. 570, 1980. |
| mouse | LC50 | inhalation | 567ppm/1H (567ppm) | BEHAVIORAL: CHANGES IN MOTOR ACTIVITY (SPECIFIC ASSAY) LUNGS, THORAX, OR RESPIRATION: CYANOSIS KIDNEY, URETER, AND BLADDER: URINE VOLUME INCREASED | Fundamental and Applied Toxicology. Vol. 1, Pg. 448, 1981. |
| mouse | LD50 | intraperitoneal | 158mg/kg (158mg/kg) | LIVER: OTHER CHANGES | Research Communications in Chemical Pathology and Pharmacology. Vol. 26, Pg. 75, 1979. |
| mouse | LD50 | oral | 171mg/kg (171mg/kg) | LIVER: OTHER CHANGES BLOOD: METHEMOGLOBINEMIA-CARBOXYHEMOGLOBIN | Research Communications in Substances Abuse. Vol. 3, Pg. 233, 1982. |
| rat | LC50 | inhalation | 420ppm/4H (420ppm) | BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) LUNGS, THORAX, OR RESPIRATION: CYANOSIS BEHAVIORAL: EXCITEMENT | Fundamental and Applied Toxicology. Vol. 8, Pg. 101, 1987. |
| rat | LD50 | oral | 83mg/kg (83mg/kg) | BEHAVIORAL: ATAXIA BEHAVIORAL: REGIDITY LUNGS, THORAX, OR RESPIRATION: DYSPNEA | JAT, Journal of Applied Toxicology. Vol. 1, Pg. 30, 1981. |