Products Categories
| CAS No.: | 7789-78-8 |
|---|---|
| Name: | Calcium hydride |
| Article Data: | 108 |
| Molecular Structure: | |
|
|
|
| Formula: | CaH2 |
| Molecular Weight: | 42.0939 |
| Synonyms: | Calcium hydride (CaH2);Calciumdihydride; |
| EINECS: | 232-189-2 |
| Density: | 1.9 |
| Melting Point: | 190 °C(lit.) |
| Boiling Point: | 1484oC |
| Solubility: | Soluble in water and alcohol. Insoluble in benzene. |
| Appearance: | greyish-white solid |
| Hazard Symbols: |
 F F
|
| Risk Codes: | 15 |
| Safety: | 24/25-43-7/8-43A |
| Transport Information: | UN 1404 4.3/PG 1 |
| PSA: | 0.00000 |
| LogP: | 0.22500 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- 90221-55-92-bromo-5-methylbenzaldehyde
- 885590-99-82,3-DIFLUORO-4-IODOBENZALDEHYDE
- 97730-31-9(S)-4'-(2-Methylbutyl)Biphenyl-4-Carbonitrile

| Conditions | Yield |
|---|---|
| In melt High Pressure; Ca melted in vac. at 350°C for 5 h, filled with H2 (5 atm) at 400°C for 5 min, at 10 atm. at 480°; elem. anal.; | 99.6% |
| In neat (no solvent) hydrogenation in a closed iron tube under pressure at temperatures between 650°C and 690°C;; | 90% |
| In neat (no solvent) hydrogenation in a closed iron tube under pressure at temperatures between 650°C and 690°C;; | 90% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating the mixture in H2-atmosphere to 420-600°C;; product-mixture;; | |
| In neat (no solvent) heating the mixture in H2-atmosphere to 420-600°C;; product-mixture;; |


| Conditions | Yield |
|---|---|
| With ammonia at red heat; | |
| With NH3 at red heat; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) on evacn. of system(metal, loaded (Ar) into reactor-autoclave of the hydrogenation apparatus)at 300-350°C (0.5-1h),cooling to room temp. and feeding hydrogen into reactor (from LaNi5 "accumulator") to give pressure of 2.8MPa, 150°C, 1h;; on gas-volumetric and chemical anal.; XRD (orthorhombic, lattice parameters); react. without an induction period and forming a clinker;; | |
| In neat (no solvent) on evacn. of system(metal, loaded (Ar) into reactor-autoclave of the hydrogenation apparatus)at 300-350°C (0.5-1h),cooling to room temp. and feeding hydrogen into reactor (from LaNi5 "accumulator") to give pressure of 3.5MPa, 200°C, 1h;; on gas-volumetric and chemical anal.; XRD (orthorhombic, lattice parameters); react. is running without an induction period and forming of a clinker;; |

| Conditions | Yield |
|---|---|
| at dark red heat; |


| Conditions | Yield |
|---|---|
| byproducts: H2, N2; vigorous reaction at 400°C; | |
| byproducts: H2, N2; vigorous reaction at 400°C; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: calcium oxide; bombarding solid CaCl2 deposited on substrate with atomar H for 60 mins in plasmochemical app. described by I. Sh. Normatov, N. Shermatov, and U. Mirsaidov, Fiz. Khim. Obrab. Mater., No. 3, 141-142 (1990);; formation of metallic layer, containing oxide phase of calcium; X-ray diffraction;; | A 0% B n/a |


| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: HCl; bombarding periodically stirred CaCl2 (on substrate) with H for 120 mins in plasmochemical app. described by I. Sh. Normatov, N. Shermatov, and U. Mirsaidov, Fiz. Khim. Obrab. Mater., No. 3, 141-142 (1990); grinding; bombarding with H for 120 mins;; X-ray diffraction, DTA, IR spectroscopy;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) reaction of CaCl2 with H2 formed by dissociation of CH4;; |


| Conditions | Yield |
|---|---|
| reversible react.; | |
| at dark red heat and at temperatures above it; |

- 109-90-0Ethane, isocyanato-
- 27206-35-51-Propanesulfonic acid,3,3'-dithiobis-, sodium salt (1:2)
- 2628-16-2Phenol, 4-ethenyl-,1-acetate
- 9007-43-6Cytochrome C
- 54048-10-1Etonogestrel
- 253265-97-3Carbonic acid,2,5-dioxo-1-pyrrolidinyl [(3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl] ester
- 65-22-54-Pyridinecarboxaldehyde,3-hydroxy-5-(hydroxymethyl)-2-methyl-, hydrochloride (1:1)
- 7397-46-8Methoxydiethylborane
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
Chemistry
IUPAC Name: Calcium hydride
Molecular Formula: CaH2
Molecular Weight: 42.093880 g/mol
Melting Point: 190 °C(lit.)
Density: 1.70 g/cm3
Sensitive: Moisture
EINECS: 232-189-2
Appearance: Greyish-white solid
Structure of Calcium hydride (CAS NO.7789-78-8):
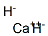
Categories of Calcium hydride (CAS NO.7789-78-8): Inorganics;Adsorbents, Filter Aids and Drying Agents;Other Drying AgentsEssential Chemicals;Materials for Hydrogen Storage;Synthetic Reagents;Metal Hydrides
Uses
Calcium hydride (CAS NO.7789-78-8) is widely used as a desiccant for basic solvents such as amines and pyridine. It is also used to pre-dry solvents prior to the use of a more reactive desiccant.
Safety Profile
Hazard Codes:F 
Risk Statements:15
15:Contact with water liberates extremely flammable gases
Safety Statements:24/25-43-7/8-43A
24/25:Avoid contact with skin and eyes
43:In case of fire, use ... (indicate in the space the precise type of fire-fighting equipment. If water increases the risk add - Never use water)
7/8:Keep container tightly closed and dry
43:In case of fire, use ... (indicate in the space the precise type of fire-fighting equipment. If water increases the risk add - Never use water)
Explosive reaction on heating with tetrahydrofuran. Mixtures with potassium chlorate and other metal oxohalogenates (e.g., chlorates; bromates; and perchlorates) are heat- and friction-sensitive explosives. Vigorous or incandescent reaction on heating with halogens (chlorine; bromine; or iodine); manganese dioxide; and silver halides (e.g., silver fluoride; silver iodide). See also CALCIUM COMPOUNDS and HYDRIDES.
Specification
Calcium hydride (CAS NO.7789-78-8) is hazardous, so the first aid measures and others should be known. Such as: When on the skin: first, should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly, get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While, it's inhaled: Remove from exposure and move to fresh air immediately. Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product: If victim is conscious and alert, give 2-4 cupfuls of milk or water. Get medical aid immediately.
In addition, you should keep it away from sources of ignition. It should be stored in a tightly closed container.



