199798-84-0 Usage
Description
BXL-628 is an analog of vitamin D3 that has diverse biological activities. It increases death of androgen-stimulated human benign prostatic hyperplasia (BPH) cells via induction of apoptosis in a dose-dependent manner. In vivo, BXL-628 completely inhibits androgen-stimulated prostate overgrowth in a rat model of BPH. It reduces expression of IL-13 and IgE/mast cell-derived protease 1 (MMCP1) and decreases edema and leukocyte infiltration in the bladder wall in a mouse model of allergen-induced interstitial cystitis. BXL-628 also reduces the number of adherent endometrial stromal cells and decreases the total weight of endometrial lesions in a mouse model of endometriosis. Formulations containing BXL-628 are under clinical investigation for the treatment of BPH.
Check Digit Verification of cas no
The CAS Registry Mumber 199798-84-0 includes 9 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 6 digits, 1,9,9,7,9 and 8 respectively; the second part has 2 digits, 8 and 4 respectively.
Calculate Digit Verification of CAS Registry Number 199798-84:
(8*1)+(7*9)+(6*9)+(5*7)+(4*9)+(3*8)+(2*8)+(1*4)=240
240 % 10 = 0
So 199798-84-0 is a valid CAS Registry Number.
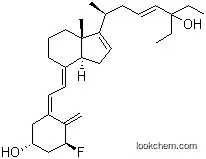
InChI:InChI=1/C29H43FO2/c1-6-29(32,7-2)17-8-10-20(3)25-14-15-26-22(11-9-16-28(25,26)5)12-13-23-18-24(31)19-27(30)21(23)4/h8,12-14,17,20,24,26-27,31-32H,4,6-7,9-11,15-16,18-19H2,1-3,5H3/b17-8+,22-12+,23-13+/t20-,24+,26-,27-,28+/m0/s1
199798-84-0Relevant articles and documents
NOVEL METHOD
-
, (2008/06/13)
There is provided a method of prevention of adhesions, eg surgical adhesions, which comprises using a vitamin D compound.
NOVEL METHOD OF TREATMENT OF MALE SUB-FERTILITY
-
Page/Page column 89-90, (2008/06/13)
-
SYNTHESIS OF 1α-FLUORO-25-HYDROXY-16-23E-DIENE-26,27-BISHOMO-20-EPI-CHOLECALCIFEROL
-
Page/Page column 74-75, (2008/06/13)
The invention provides a method of producing 20-methyl vitamin D3 compounds of formula (I). The method includes allylic and olefin oxidation, decarbonylation, carbonyl reduction, fluoride substitution, epoxide deoxygenation, and Wittig-type couplings.