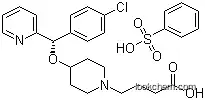
190786-44-8 Usage
Description
Different sources of media describe the Description of 190786-44-8 differently. You can refer to the following data:
1. Betotastine was introduced in Japan for the treatment of allergic rhinitis. This
structurally-related derivative of chlorpheniramine and ebastine is prepared by
condensation of optically-resolved 4-[1-(4-chlorophenyl)-1-(2-pyridyl)-methoxy]piperidine
with ethyl 4-bromobutyrate followed by ester hydrolysis. Betotastine is the seventh
marketed non-sedating histamine H1 antagonist. Its very low sedative side effect is due to
very poor penetration in the central nervous system. Besides its potent and long-acting
activity in models of allergic rhinitis, betotastine was also shown to act as a PAF antagonist and inhibit LTD4 in tracheal smooth muscle and ileum, IL-5 production by human peripheral
blood mononuclear cells as well as eosinophil infiltration in the airway and peripheral
blood. As a consequence, it is currently being developed against other allergic and
respiratory disorders.
2. Bepotastine is an antagonist of the histamine H1 receptor that is selective over H3, α1-, α2-, and β-adrenergic, dopamine D2long, serotonin 5-HT2, muscarinic acetylcholine, and benzodiazepine receptors. It reduces dye leakage from the nasal passages of rats acutely sensitized to an antigen (ED50 = 0.03 mg/kg) and inhibits histamine-induced bronchoconstriction in the anesthetized dog (ED50 = 3.2 μg/kg). Bepotastine prevents conjunctival vascular hyperpermeability in a guinea pig model of conjunctivitis in a dose-dependent manner. Formulations containing bepotastine have been used in the treatment of itching associated with allergic conjunctivitis.
Chemical Properties
Off-White to Light Beige Solid
Originator
UBE (Japan)
Uses
Different sources of media describe the Uses of 190786-44-8 differently. You can refer to the following data:
1. Bepotastine is a non-sedating, selective antagonist of histamine 1 (H1) receptor with pIC50 of 5.7
2. Bepotastine is a histamine H1 receptor anatagonist. Bepotastine suppresses some allergic inflammatory processes such as allergic rhinitis, chronic urticaria or pruritus associated with skin conditions
(eczema/dermatitis, prurigo or pruritus cutaneus).
Definition
ChEBI: An organosulfonate salt obtained by combining equimolar amounts of bepotastine and benzenesulfonic acid. A topical, selective and non-sedating histamine (H1) receptor antagonist used for treatment of itching associated with allergic co
junctivitis.
Manufacturing Process
Manufacturing process for BEPOTASTINE BESILATE includes these steps as follows: Step A: Synthesis of Methyl 2-endo-hydroxy-1-exo-hydroxymethyl-3a,8b-cis-2,3,3a,8b-tetrahydro-
1H-5-cyclopenta[b]benzofurancarboxylate,Step B: Synthesis of Methyl 3-methyl-trans-4a-cisoid-4a,5a-cis-5a-1,4a,5,5a,10b,10c-hexahydro-7-
dioxin o[5,4-a]cyclopenta[b]benzofurancarboxylate,Step C: Synthesis of 3-Methyl-trans-4a-cosoid-4a,5a-cis-5a-1,4a,5,5a,10b,10c-hexahydro-7-
dioxino[5,4-a]cyclopenta[b]benzofuranylmethanol,Step D: Synthesis of 7-Chloromethyl-3-methyl-trans-4a-cisoid-4a,5a-cis-5a-1,4a,5,5a,10b,10c�hexahydrodioxino[5,4-a]cyclopenta[b]benzofuran,Step E: Synthesis of 4-[3-Methyl-trans-4a-cisoid-4a,5a-cis-5a-1,4a,5,5a,10b,10c-hexahydro-7-
dioxino[5,4-a]cyclopenta[b]benzofuranyl]butyric acid, Step F: Synthesis of Methyl 4-[2-endo-hydroxy-1-exo-hydroxymethyl-3a,8b-cis-2,3,3a,8b�tetrahydro-1H-5- cyclopenta[b]benzofuranyl]butyrate, Step G: Synthesis of Methyl 4-[2-endo-acetoxy-1-exo-hydroxymethyl-3a,8b-cis-2,3,3a,8b�tetrahydro-1H-5-cyclopenta[b]benzofuranyl]butyrate, Step H: Synthesis of Methyl ester of 11,15-dideoxy-11-acetoxy-16-methyl-15-oxo-18,19-
tetradehydro-5,6,7-trinor-4,8-inter-m-phenylene PGI2,Step I: Synthesis of 11-Deoxy-11-acetoxy-16-methyl-18,19-tetradehydro-5,6,7-trinor-4,8-inter-m�phenylene PGI2.To a solution of 54 mg of methyl ester of 11-deoxy-11-acetoxy-16-methyl-
18,19-tetradehydro-5,6,7-trinor-4,8-inter-m-phenylene PGI2 in 4.5 ml of
anhydrous methanol was added 0.001 ml of 4.8 N sodium methoxide under
argon, and the reaction mixture was stirred for 1.5 hours at room
temperature.
After addition of acetic acid to the reaction mixture and concentration of the
mixture, the residue was dissolved in 20 ml of ethyl acetate, and the solution
was washed with aqueous saturated solution of sodium hydrogen carbonate,
water and aqueous saturated solution of sodium chloride, dried and
concentrated to afford 55 mg of an oily material.
This oily material was purified by column chromatography using ethyl acetate
and cyclohexane (3:1) as eluent to give 48 mg of the methyl ester of 16-
methyl-18,19-tetradehydro-5,6,7-trinor-4,8-inter-m-phenylene PGI2.
Brand name
Talion
Therapeutic Function
Antiallergic
Check Digit Verification of cas no
The CAS Registry Mumber 190786-44-8 includes 9 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 6 digits, 1,9,0,7,8 and 6 respectively; the second part has 2 digits, 4 and 4 respectively.
Calculate Digit Verification of CAS Registry Number 190786-44:
(8*1)+(7*9)+(6*0)+(5*7)+(4*8)+(3*6)+(2*4)+(1*4)=168
168 % 10 = 8
So 190786-44-8 is a valid CAS Registry Number.
InChI:InChI=1/C21H25ClN2O3.C6H6O3S/c22-17-8-6-16(7-9-17)21(19-4-1-2-12-23-19)27-18-10-14-24(15-11-18)13-3-5-20(25)26;7-10(8,9)6-4-2-1-3-5-6/h1-2,4,6-9,12,18,21H,3,5,10-11,13-15H2,(H,25,26);1-5H,(H,7,8,9)/t21-;/m1./s1
190786-44-8Relevant articles and documents
Improved Synthesis of Bepotastine Besilate
Han,Xia,Sun,Zou
, p. 206 - 210 (2021/03/15)
-
IMPROVED PROCESS FOR THE MANUFACTURE OF BEPOTASTINE AND ITS BESILATE SALT
-
, (2019/05/02)
The present invention discloses a process for preparation of Bepotastine and its Besilate salt of formula I with good yield and purity. The invention also describes a process for recycle and reuse of the Ethyl-4-hydroxy piperidine-1-carboxylate from the aqueous medium after isolating the 2-[(S)-(4-Chlorophenyl)(piperidin-4- yloxy)methyl]pyridine, for subsequent batches in the production of Ethyl 4-[(4- Chlorophenyl)(pyridin-2-yl)methoxy]piperidine-1-carboxylate. The invention further discloses novel intermediates, viz., 2-[Chloro(4- chlorophenyl)methyl]pyridine hydrochloride and bis{2-[(S)-(4- Chlorophenyl)(piperidin-4-yloxy)methyl]pyridine} Dibenzoyl tartrate, useful in the preparation of Bepotastine and its Besilate salt.
Iridium-Catalyzed Highly Enantioselective Transfer Hydrogenation of Aryl N-Heteroaryl Ketones with N-Oxide as a Removable ortho-Substituent
Liu, Qixing,Wang, Chunqin,Zhou, Haifeng,Wang, Baigui,Lv, Jinliang,Cao, Lu,Fu, Yigang
, p. 971 - 974 (2018/02/23)
A highly enantioselective transfer hydrogenation of non-ortho-substituted aryl N-heteroaryl ketones, using readily available chiral diamine-derived iridium complex (S,S)-1f as a catalyst and sodium formate as a hydrogen source in a mixture of H2O/i-PrOH (v/v = 1:1) under ambient conditions, is described. The chiral aryl N-heteroaryl methanols were obtained with up to 98.2% ee by introducing an N-oxide as a removable ortho-substituent. In contrast, no more than 15.1% ee was observed in the absence of an N-oxide moiety. Furthermore, the practical utility of this protocol was also demonstrated by gram-scale asymmetric synthesis of bepotastine besilate in 51% total yield and 99.9% ee.