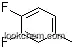
2927-34-6Relevant articles and documents
Two-step regioselective synthesis of 1,2-difluorobenzenes from chlorotrifluoroethylene and buta-l,3-dienes
Lipkind, M. B.,Nefedov, O. M.,Volchkov, N. V.
, p. 68 - 75 (2020/04/21)
The gas-phase copyrolysis of chlorotrifluoroethylene with buta-1,3-diene, penta-1,3-diene, or isoprene in a flow reactor at 440–480°C gave 4-chloro-4,5,5-trifluorocyclohex-1-enes. The latter treated with aqueous KOH under condition of phase-transfer catalysis were selectively converted into 1,2-difluorobenzene, 2,3-difluorotoluene, or 3,4-difluorotoluene.
Friedel–Crafts Type Methylation with Dimethylhalonium Salts
H?mmerling, Sebastian,Vo?nacker, Patrick,Steinhauer, Simon,Beckers, Helmut,Riedel, Sebastian
, p. 14377 - 14384 (2020/10/12)
The dimethylchloronium salt [Me2Cl][Al(OTeF5)4] is used to methylate electron-deficient aromatic systems in Friedel–Crafts type reactions as shown by the synthesis of N-methylated cations, such as [MeNC5F5]+, [MeNC5F4I]+, and [MeN3C3F3]+. To gain a better understanding of such fundamental Friedel–Crafts reactions, the role of the dimethylchloronium cation has been evaluated by quantum-chemical calculations.
Method of preparing fluoroaromatic compounds
-
, (2008/06/13)
The present invention provides a method of preparing ortho-difluorobenzene derivatives, which comprises (a) providing a mixture of cyclohexenes by reacting chlorotrifluoroethylene (CTFE) and 1,3-diene in a flow reactor and distilling the resultant, and (b) dehydrohalogenating the mixture of cyclohexenes with a phase transition catalyst in the presence of alkali metal hydroxide at temperature range of 40 to 150 °C without using any organic solvent. The distillate having low boiling point, which is obtained during distillation of the resultant, is recycled into the flow reactor. The present invention also provides a method of preparing 2-chloro-4,5-difluorobenzoic acid, which comprises (a) providing a mixture of 4-chloro-1-methyl-4,5,5-trifluorocyclohexene and 5-chloro-1-methyl-4,4,5-trifluorocyclohexene by reacting chlorotrifluoroethylene (CTFE) and isoprene and distilling the resultant, (b) dehydrohalogenating said mixture in the presence of alkali metal hydroxide and a phase transition catalyst to form 3,4-difluorotoluene, (c) reacting said 3,4-difluorotoluene with chlorine gas without using any organic solvent to form 2-chloro-4,5-difluorotoluene, (d) photo-reacting said 2-chloro-4,5-difluorotoluene with chlorine gas under a lighting mercury lamp without using any organic solvent to form 2-chloro-4,5-difluorobenzotrichloride, and (e) reacting said 2-chloro-4,5-difluorobenzotrichloride with aqueous acid solution without using any organic solvent. The present invention also provides a method of preparing 2-chloro-4,5-difluorobenzoyl chloride by reacting the 2-chloro-4,5-difluorobenzotrichloride of step (e) above with zinc oxide.