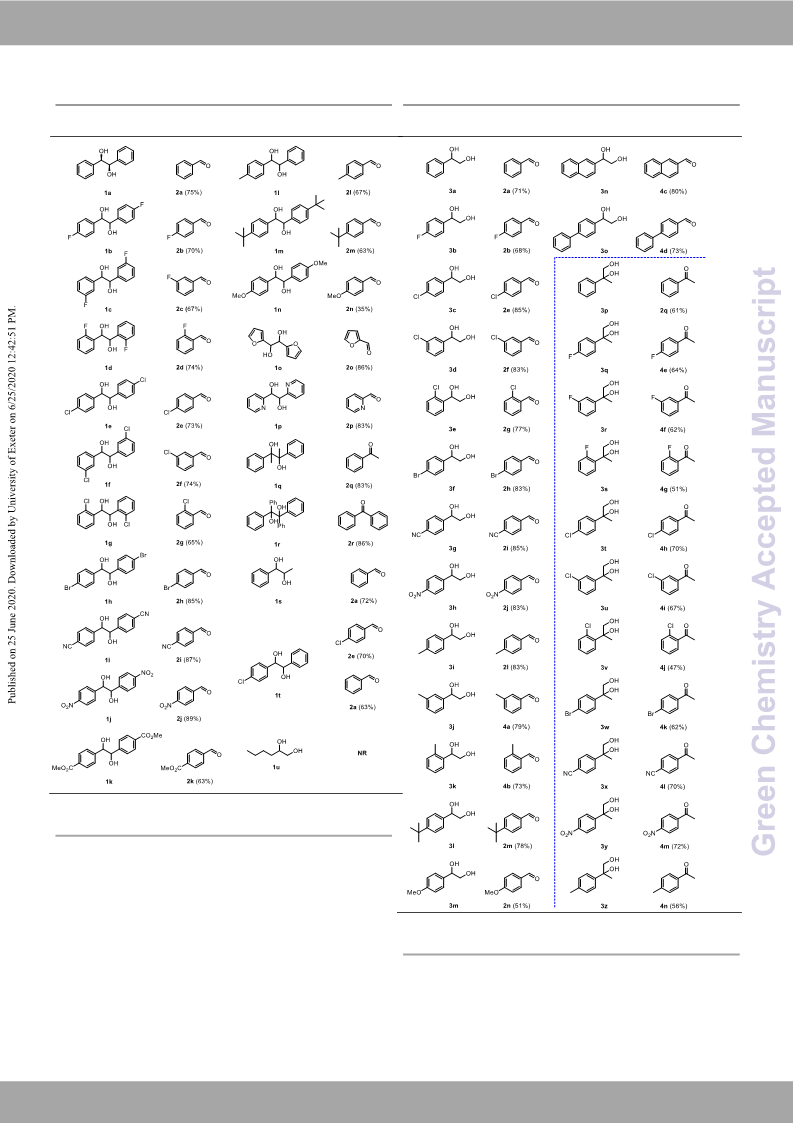
Please Gd roe en no Ct ha ed mj u iss tt r my argins
Page 6 of 8
ARTICLE
Journal Name
9
separated aqueous medium containing CN620 could be
recycled directly for 10 runs with the products in HPLC yields
from 74 to 85% (Figure 2).
Y. Ema, M. Torii, Y. Ishizuka and I. Souta, Tetrahedron Lett.,
DOI: 10.1039/D0GC01727B
2
010, 51, 3619-3622; (b) E. Amadio, J. González-Fabra, D.
Carraro, W. Denis, B. Gjoka, C. Zonta, K. Bartik, F. Cavani, S.
Solmi, C. Bo and G. Licini, Adv. Synth. Catal., 2018, 360, 3286-
3
296.
Conclusions
10 (a) N. García, R. Rubio-Presa, P. García-García, M. A.
Fernández-Rodríguez, M. R. Pedrosa, F. J. Arnáiz and R. Sanz,
Green Chem., 2016, 18, 2335-2340; (b) A. M. Khenkin and R.
Neumann, Adv. Synth. Catal., 2002, 344, 1017-1021.
In summary, an environmentally-friendly and effective
metal-free aqueous photocatalytic C-C bond oxidative cleavage
of vicinal diols has been developed using CN620 as a recyclable 11 H. Noack, V. Georgiev, M. R. Blomberg, P. E. Siegbahn and A.
J. Johansson, Inorg Chem., 2011, 50, 1194-1202.
photocatalyst and CTAB as the surfactant. A series of vicinal
diols could be efficiently cleaved and converted into the
corresponding aldehydes or ketones. TEM revealed that the
micelle is probably on the surface of the catalyst. Preliminary
mechanism revealed that both the superoxide radicals and
valence band holes played an important role in the reaction
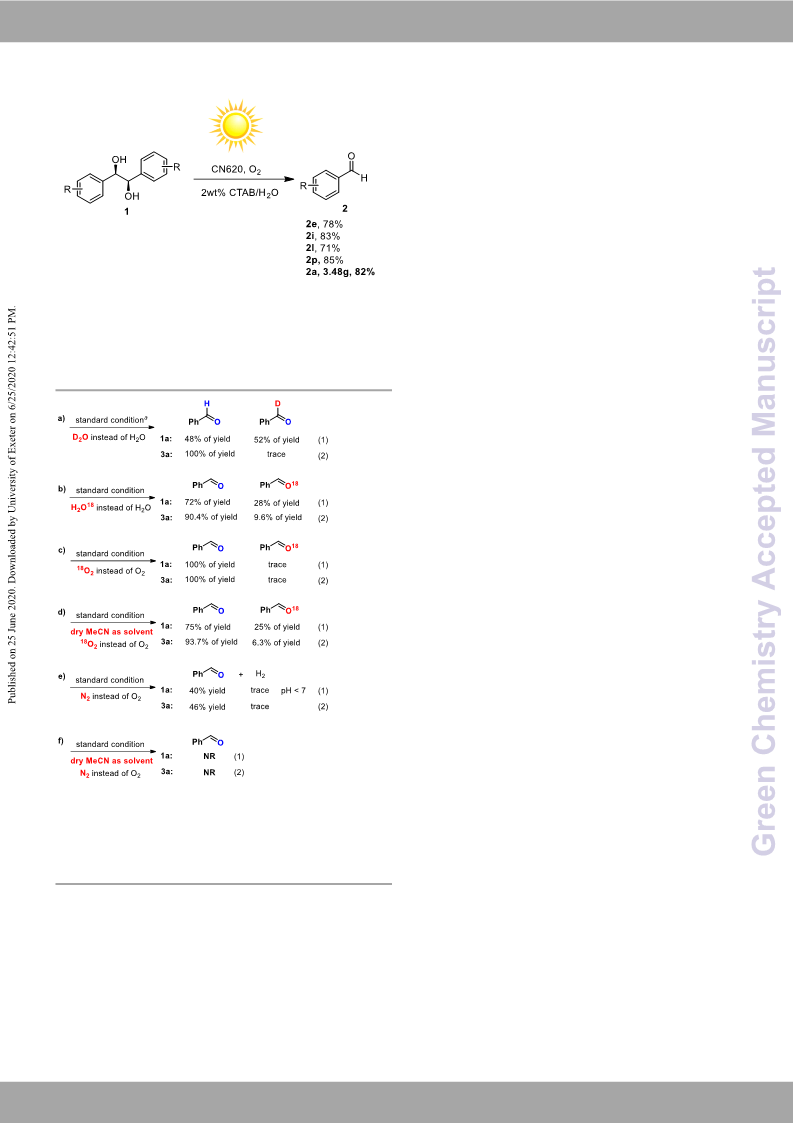
process. Further isotope experiment confirmed β-scission/HAT
process and oxidation/hydrolysis/dehydration process in the
reaction which is different from previous reports. Notably, the
CN620/micellar catalyst system could be recycled and reused 10
times with satisfactory yield. Moreover, this reaction could be
carried out under solar light irradiation and is applicable for
large-scale reactions. These features make this reaction a
potential green and sustainable approach for both academic
and industrial applications.
1
1
2 L. Prati and M. Rossi, J. Mol. Catal. A: Chem., 1996, 110, 221–
2
26.
3 (a) E. Santacesaria, A. Sorrentino, F. Rainone, M. Di Serio and
F. Speranza, Ind. Eng. Chem. Res., 2000, 39, 2766-2771; (b) P.
Mastrorilli, G. P. Suranna, C. F. Nobile, G. Farinola and L. Lopez,
J. Mol. Catal., 2000, 156, 279–281; (c) H. Luo, L. Wang, S.
Shang, J. Niu and S. Gao, Commun. Chem., 2019,
4 (a) X. Lang, J. Zhao and X. Chen, Chem. Soc. Rev., 2016, 45
026-3038; (b) M. H. Shaw, J. Twilton and D. W. MacMillan, J.
2, 17.
1
,
3
Org. Chem., 2016, 81, 6898-6926; (c) J. Xie, H. Jin and A. S. K.
Hashmi, Chem. Soc. Rev., 2017, 46, 5193-5203; (d) W. Ding, L.
Q. Lu, Q. Q. Zhou, Y. Wei, J. R. Chen and W. J. Xiao, J. Am. Chem.
Soc., 2017, 139, 63-66; (e) A. Sagadevan, V. P. Charpe, A.
Ragupathi and K. C. Hwang, J. Am. Chem. Soc., 2017, 139
896-2899; (f) K. Wang, L.-G. Meng, Q. Zhang and L. Wang,
Green Chem., 2016, 18, 2864-2870; (g) S. K. Pagire, P.
Kreitmeier and O. Reiser, Angew. Chem., Int. Ed., 2017, 56
0928-10932; (h) Y. Ding, W. Zhang, H. Li, Y. Meng, T. Zhang,
Q.-Y. Chen and C. Zhu, Green Chem., 2017, 19, 2941-2944; (i)
,
2
,
1
L. Zhang, H. Yi, J. Wang and A. Lei, Green Chem., 2016, 18
,
Conflicts of interest
There are no conflicts to declare.
5
122-5126; (j) L. Ren, M.-M. Yang, C.-H. Tung, L.-Z. Wu and H.
Cong, ACS Catal., 2017,
7
, 8134-8138; (k) B. Muhldorf and R.
Wolf, Angew. Chem., Int. Ed., 2016, 55, 427-430; (l) T. F. Niu,
D. Y. Jiang, S. Y. Li, B. Q. Ni and L. Wang, Chem. Commun., 2016,
5
2
, 13105-13108.
Acknowledgements
1
1
5 (a) C. K. Prier, D. A. Rankic and D. W. MacMillan, Chem. Rev.,
2
013, 113, 5322-5363; (b) Y. Xi, H. Yi and A. Lei, Org. Biomol.
The authors gratefully acknowledge the National Natural
Science Foundation of China (no.21808085), China Postdoctoral
Science Foundation (2018M630519) and Postdoctoral Science
Foundation of Jiangsu Province, China (1701096B).
Chem., 2013, 11, 2387-2403.
6 (a) N. A. Romero and D. A. Nicewicz, Chem. Rev., 2016, 116
1
2
,
0075-10166; (b) D. P. Hari and B. Kꢀnig, Chem. Commun.,
014, 50, 6688-6699.
1
1
7 J. Schwarz and B. Kꢀnig, Chem. Commun., 2019, 55, 486-488.
8 (a) W. J. Ong, L. L. Tan, Y. H. Ng, S. T. Yong and S. P. Chai, Chem.
Rev., 2016, 116, 7159-7329; (b) D. Masih, Y. Ma and S. Rohani,
Appl. Catal., B., 2017, 206, 556-588; (c) J. Wen, J. Xie, X. Chen
and X. Li, Appl. Surf. Sci., 2017, 391, 72-123; (d) Z. Zhou, Y.
Notes and references
1
A. S. Perlin, Advances in Carbohydrate Chemistry and Bio-
chemistry, Academic Press, Cambridge, 2006.
Zhang, Y. Shen, S. Liu and Y. Zhang, Chem. Soc. Rev., 2018, 47
298-2321.
,
2
3
R. Criegee, Ber. Dtsch. Chem. Ges., 1931, 64, 260–266.
2
(a) L. Malaprade, Bull. Soc. Chim. Fr., 1934, 5, 833-852; (b) L.
1
9 (a) A. Savateev, I. Ghosh, B. Kꢀnig and M. Antonietti, Angew.
Chem., Int. Ed., 2018, 57, 15936-15947; (b) A. Savateev and M.
Malaprade, C. R. Hebd, Seances Acad. Sci. Roum., 1928, 186
,
3
6
82–385; (c) L. Malaprade. Bull. Soc. Chim. Fr., 1928, 43, 683-
96.
Antonietti, ACS Catal., 2018, 8, 9790-9808; (c) X. Sun, X. Luo,
X. Zhang, J. Xie, S. Jin, H. Wang, X. Zheng, X. Wu and Y. Xie, J.
Am. Chem. Soc., 2019, 141, 3797-3801; (d) B. Kurpil, K. Otte,
A. Mishchenko, P. Lamagni, W. Lipinski, N. Lock, M. Antonietti
and A. Savateev, Nat. Commun., 2019, 10, 945; (e) B. Pieber,
J. A. Malik, C. Cavedon, S. Gisbertz, A. Savateev, D. Cruz, T. Heil,
4
5
6
N. Obara, S. Hirasawa, M. Tamura, Y. Nakagawa and K.
Tomishige, ChemCatChem., 2016, , 1732-1738.
T. R. Felthouse, P. B. Fraundorf, R. M. Friedman and C. L.
Schosser, J. Catal., 1991, 127, 393-420.
S. Solmi, E. Rozhko, A. Malmusi, T. Tabanelli, S. Albonetti, F.
Basile, S. Agnoli and F. Cavani, Appl. Catal., A., 2018, 557, 89-
9
8
G. Zhang and P. H. Seeberger, Angew. Chem. Int. Ed., 2019, 58
,
9
575-9580; (f) Y. Xiao, G. Tian, W. Li, Y. Xie, B. Jiang, C. Tian, D.
8. (b) E. akezawa, S. Sakaguchi and Y. Ishii, Org. Lett., 1999,
, 713–715.
Zhao and H. Fu, J. Am. Chem. Soc., 2019, 141, 2508-2515; (g)
I. Camussi, B. Mannucci, A. Speltini, A. Profumo, C. Milanese,
L. Malavasi and P. Quadrelli, ACS Sustainable Chem. Eng.,
1
7
8
Z. Z. Zhou, M. Liu, L. Lv and C. J. Li, Angew. Chem., Int. Ed.,
2
018, 57, 2616-2620.
2
019,
X. Wang, ACS Catal., 2019,
Markushyna and A. Savateev, ACS Catal., 2019,
7
, 8176-8182; (h) H. Ou, C. Tang, X. Chen, M. Zhou and
, 2949-2955; (i) B. Kurpil, Y.
, 1531-1538;
(a) V. Escande, C. H. Lam, P. Coish and P. T. Anastas, Angew.
Chem., Int. Ed., 2017, 56, 9561-9565; (b) S.-S. Meng, L.-R. Lin,
X. Luo, H.-J. Lv, J.-L. Zhao and A. S. C. Chan, Green Chem., 2019,
9
9
(
j) C. Cavedon, A. Madani, P. H. Seeberger and B. Pieber, Org.
21, 6187-6193.
Lett., 2019, 21, 5331-5334; (k) I. Ghosh, J. Khamrai, A.
6
| J. Name., 2012, 00, 1-3
This journal is © The Royal Society of Chemistry 20xx
Please do not adjust margins

 Chen, Jiayang
Chen, Jiayang
 Chen, Shengjun
Chen, Shengjun
 Dong, Xinyu
Dong, Xinyu
 Hong, Mei
Hong, Mei
 Ni, Bangqing
Ni, Bangqing
 Niu, Tengfei
Niu, Tengfei
 Zhang, Tianhao
Zhang, Tianhao