IR vibration bands of the unreacted solid in the 1800È1200
cm~1 region are replaced in the reacted solid (prior to solvent
extraction) by low intensity bands at 1272 and 1700 cm~1,
hundred milligrams of ZnCrÈAc É 3H O (ZnCrÈBn É 3H O)
2
2
were mixed with 29.9 ll (25.9 ll) of benzyl bromide or with
43.4 ll (37.6 ll) of 1-bromooctane. The reaction took place by
heating either in a domestic MW oven (600 W power) or in a
conventional oven at 100 ¡C. After the reaction was com-
pleted, the resulting products were extracted with methanol
and analysed by GC-MS. The results of the alkylation of the
intercalated carboxylates, after 15 min of MW treatment or
5 h of heating at 100 ¡C, are given in Table 1.
which correspond to l
and l
vibrations, indicating the
cho
c/o
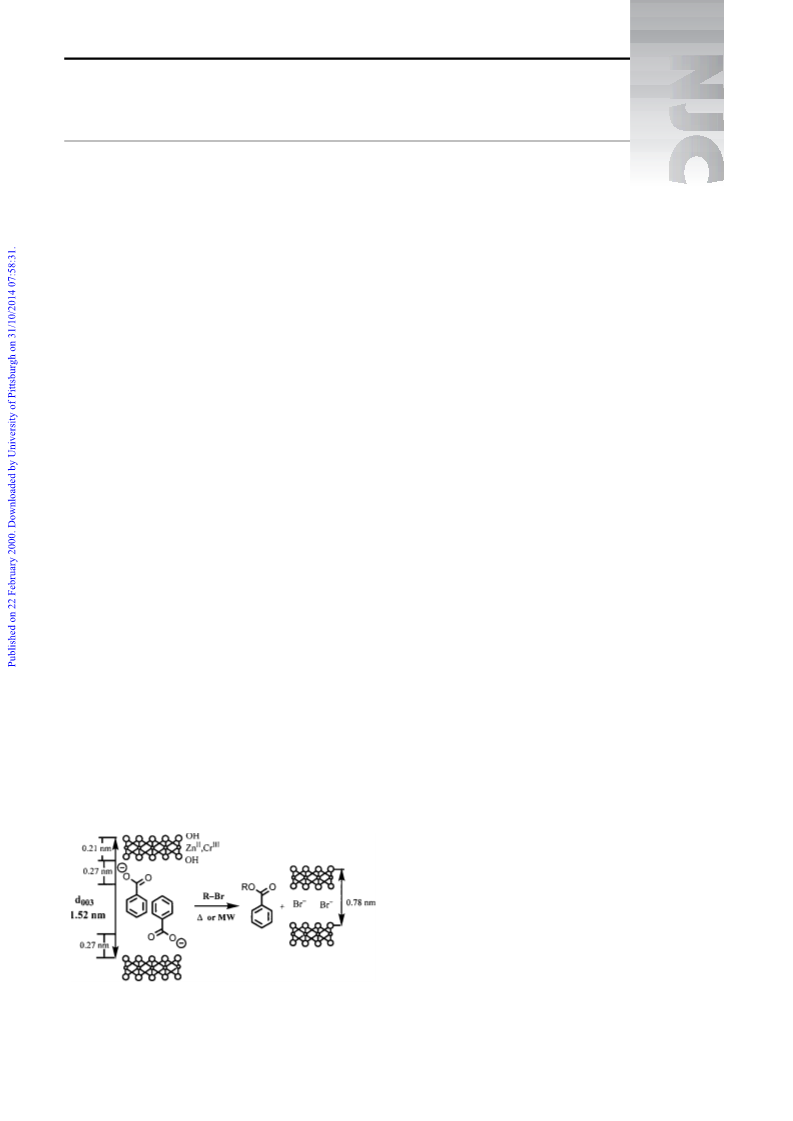
formation of the ester in the selected example (Fig. 2).
Regarding the reaction mechanism, it should be pointed out
that the low dipolar moment of the organic halides used, as
well as the limited LDH swelling capacity, are factors that
determine the controlled entry of these reagents into the inter-
layer region of the solid where the active anions are located.
We assume that this topotactic reaction takes place via di†u-
sion of anions from the layer edges towards the intracrystal-
line space of the LDH solid, as proposed by Martin and
Pinnavaia16 in the nucleophilic substitution reactions of
halogen atoms of organic halides reacting with LDH
exchanged by halides. In conclusion, although supported reac-
tions between carboxylates and alkyl and aryl halides were
previously studied,1,2 in the present work we report the reacti-
vity of a solid material (an LDH) that contains carboxylates,
selected here as a model example. According to the described
general procedure, the nature of the anionic species
(carboxylates in this case) involved in the reactions can be
extended to other anions, in particular to unstable or toxic
compounds in their anionic form. The processes reported here
are topotactic reactions, which are completely di†erent from
the ““conventionalÏÏ procedures to activate anions on sup-
ported reagents such as alumina, but they give similar good
yields. It will now be possible to conduct reactions involving
anions in a constrained region, which could change the selec-
tivity of the resulting products as reported for processes
occurring in the interior of other layered or microporous
solids.17 After reaction, it is possible to recover the solid, con-
taining the other anion (bromide in our case) and, separately,
the product of the reaction, the O-alkylated carboxylate com-
pounds in these preliminary reported examples.
Acknowledgements
This work was partially supported by the CICYT, Spain. The
authors gratefully acknowledge technical assistance from J.
Merino and M. A. Muro. We also thank M. A. Martin
Luengo and P. Aranda for helpful discussions and M. Yates
for revising the manuscript.
Notes and references
1
(a) G. Bram, T. FillebeenÈKhan and N. Geraghty, Synth.
Commun., 1980, 10, 279; (b) P. Lazlo, Preparative Chemistry using
Supported Reagents, Academic Press, San Diego, 1987.
(a) E. Gutierrez, A. Loupy, G. Bram and E. RuizÈHitzky, T etra-
hedron L ett., 1989, 30, 945; (b) G. Bram, A. Loupy, M. Majdoub,
E. Gutierrez and E. RuizÈHitzky, T etrahedron, 1990, 46, 5167.
R. S. Varma, Green Chem., 1999, 1, 43.
2
3
4
E. M. Sabbar, M. E. Deroy, A. Ennaqadi, C. Gueho and J. P.
Besse, Chem. Mater., 1998, 10, 3856.
5
(a) D. M. P. Mingos and D. R. Baghurst, Chem. Soc. Rev., 1991,
20, 1; (b) Microwave Enhanced Chemistry. Fundamentals, Sample
Preparation and Applications, ed. H. M. Kingston and J. Haswell,
American Chemical Society, Washington, D.C., 1997.
K. A. Carrado and A. Kostapapas, Solid State Ionics, 1988, 26,
77.
6
7
8
A. Vaccari, Catal. T oday, 1998, 41, 53.
F. Medina, D. Tichit, B. Coq, A. Vaccari and N. T. Dung, J.
Catal., 1997, 167, 142.
Further work is now in progress in order to investigate the
inÑuence of the nature of the cations located in the octahedral
sheets of the LDH matrices, as well as the role of the water
molecules in the studied reactions.
9
S. Velu and C. S. Swamy, Appl. Catal., 1996, 145, 141.
10 E. Suzuki, M. Okamoto and Y. Ono, Chem. L ett., 1989, 1485.
11 M. Meyn, K. Beneke and G. Lagaly, Inorg. Chem., 1990, 29, 5201.
12 A. DeRoy, C. Forano, K. El-Malki and J. P. Besse, in Expanded
Clays and Other Microporous Materials, ed. M. L. Ocelli and H.
F. Robinson, Van Norstand Reinhold, New York, 1992.
13 H. P. Boehm, J. Steinle and C. Vieweger, Angew. Chem., Int. Ed.
Engl., 1977, 16, 265.
14 B. Dietrich, P. Viout and J. M. Lehn, Macrocyclic Chemistry.
Aspects of Organic and Inorganic Supramolecular Chemistry,
VCH, Weinhem, 1993.
Experimental
The preparation of [Zn Cr(OH) ]`[NO ~] É nH O was
2
6
3
2
carried out as described in the literature.11h13 The intercalated
LDH were obtained by anionic exchange: 1 g of ZnCrÈNO
precursor was dispersed into a 1 M solution of the corre-
15 M. MartinezÈLara, A. JimenezÈLopez, L. MorenoÈLeal, S.
Bruque, B. Casal and E. RuizÈHitzky, Mater. Res. Bull., 1985, 20,
549.
3
sponding sodium carboxylate (acetate or benzoate). The reac-
tion mixture was continuously stirred for 24 h at room
temperature under a nitrogen Ñow. Then the precipitate was
recovered by centrifugation and the resulting solid washed
three times with deionized water and, Ðnally, dried at room
temperature.
These intercalation compounds were impregnated with a
stoichiometric amount of halide, either 1-bromooctane or
benzyl bromide, and placed in a TeÑon} reactor. One
16 K. J. Martin and T. J. Pinnavaia, J. Am. Chem. Soc., 1986, 108,
541.
17 (a) Chemical Reactions in Organic and Inorganic Constrained
Systems, ed. R. Setton, NATO ASI Series, Series C: Mathemati-
cal & Physical Sciences, vol. 165, Reydel Pub. Co., Dordrecht,
1986; (b) J. M. Thomas and W. J. Thomas, Principles and Prac-
tice of Heterogeneous Catalysis, VCH, Weinheim, 1997.
L etter a909263c
New J. Chem., 2000, 24, 119È121
121

 Garcia-Ponce, Angel Luis
Garcia-Ponce, Angel Luis
 Prevot, Vanessa
Prevot, Vanessa
 Casal, Blanca
Casal, Blanca
 Ruiz-Hitzky, Eduardo
Ruiz-Hitzky, Eduardo