1
2672 J. Phys. Chem. A, Vol. 114, No. 48, 2010
Coleman and Ault
-1
at 880 cm . Finally, these unimolecular stabilization pathways
have activation energies calculated to range from approximately
-1 26
1
0 to 20 kcal mol . Thus, these pathways are much less likely
and do not appear to play a role under the present experimental
conditions.
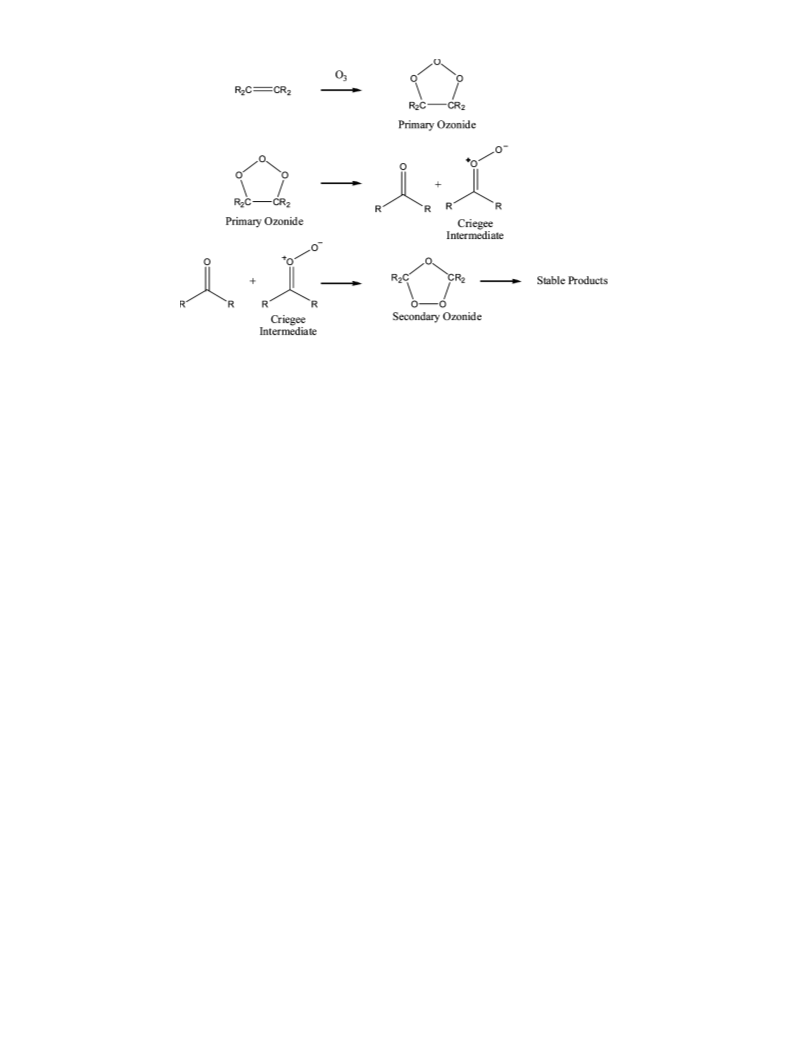
Finally, the Criegee mechanism predicts the formation of the
CI along with acetone from the decomposition of the POZ of
DMB. In view of the definitive observation of acetone (see
above), the very good agreement of calculation and experiments
Figure 5. Scheme for the O atom attack on the double bond of DMB.
for the CdO-O stretch mode and, overall, the observation of
an isotopic quartet in the scrambled 1
6,18
3
O experiments, and
that the O atom will find a substrate molecule and react. Most
of the observed product absorptions that grew in when irradiated
from λ g 220 to g580 nm can be assigned to either acetone,
tetramethyloxirane, or 3,3-dimethyl-2-butanone based on lit-
the lack of observation of anticipated rearrangement products
of the CI, the eVidence collectiVely supports assignment of the
80 cm-1 band to the CI of DMB. Further, this provides
8
definitive evidence that tetra-substituted alkenes follow the
Criegee mechanism.
2
3,29
erature spectra and theoretical calculations.
-
1
It is noteworthy that the 880 cm band assigned to the CI
Earlier studies have suggested that formation of a SOZ from
the CI of tetra-alkyl-substituted alkenes would be more difficult
was reduced in intensity when irradiated with light with λ <
5
50 nm. At the same time, several new absorptions were
1
9
than that from a lesser substituted alkene. Later studies have
disputed this suggestion. While the SOZ of DMB has not been
reported in the gas phase or in inert matrixes, one study reported
the observation of this species immobilized in a polyethylene
observed when irradiating in this wavelength range. These
included two weak carbonyl stretch absorptions at 1733 and
-1
-1
1
756 cm and a broad O-H stretch at 3431 cm . These
observations suggest destruction of the CI and formation of
related products. As discussed above, unimolecular rearrange-
ment products, including hydroxyacetone, dioxirane, and methyl
acetate, are likely candidates. In this case, the energy required
27
film. However, this study lacked isotopic labeling and theoreti-
cal calculations, and the broad spectral features made this
identification tentative. In the present study, additional weak
-1
product absorptions were observed at 1206, 870, and 829 cm .
Incorporation of 18O labeling and comparison to theoretical
calculations for the SOZ provide good evidence that these bands
can be assigned to the SOZ of DMB. In particular, the most
-1
for the CI to overcome the calculated 10-20 kcal mol barrier
leading to these stable compounds is available through absorp-
tion of light. Sander found that while generating carbonyl oxides
(
CIs) via another route, irradiation at select wavelengths led to
-
1
intense calculated absorption for the SOZ came at 1223 cm
different products (λ ) 515 nm leads to dioxirane formation
with a -4 cm- calculated O shift without scaling. This
1
18
30
and further to an ester at λ ) 438 nm). In similar manner, the
corresponds nicely to the most intense experimental absorption
-1
bands observed here at 1733, 1756, and 3431 cm are assigned
for the SOZ at 1206 cm- that had an O shift of -4 cm .
1
18
-1
to hydroxyacetone and methyl acetate. In addition, there are
Similar agreement of experiment and theory was seen for the
-1
three weak bands at 900, 1022, and 1330 cm assigned to the
8
29 and 870 cm- bands, as shown in Table 1. While many
1
31
dioxirane that serves as an intermediate between the CI and
more bands are anticipated for the SOZ, additional absorptions
were not observed in part due to overlap with parent bands in
the reaction spectrum and in part due to low intensities. No
-1
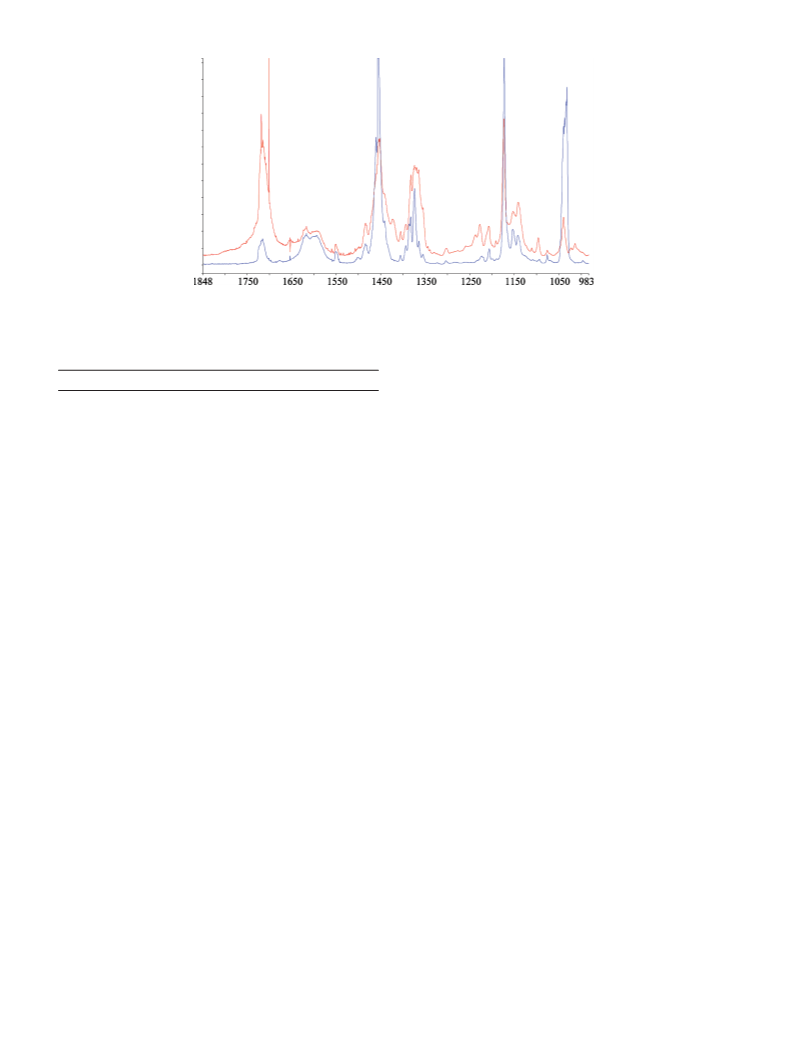
methyl acetate. In Figure 6, the CI band at 880 cm decreases
-1
as the dioxirane band at 900 cm increases. These results
present direct eVidence of the waVelength dependence of the
photodestruction of a CI.
additional bands were observed for the SOZ in the 16,18
3
O mixed
isotope experiment. Considering that there are eight calculated
isotopomers for each SOZ band, the intensities of each
individual band in the experimental study would be greatly
reduced to the point that they would not be observed. Table 3
shows calculated positions for the SOZ isotopomers.
O + DMB Merged Jet. In merged jet deposition, the mixing
3
of ozone and DMB occurred at room temperature in a flow tube
outside of the matrix cell. Additional reaction time is available
in this mode compared to that in twin jet and has led to “late”
thermal reaction products in previous studies. In these experi-
ments, bands of the precursors and early intermediate species
decreased significantly compared to those in twin jet spectra,
and a number of new product absorptions were observed. This
indicates that extensive reaction occurred during the transit
through the merged jet or reaction region. In particular, there
were numerous product bands in the carbonyl stretching region
O + DMB Twin Jet Photochemistry. In twin jet argon
3
matrix experiments that were irradiated with light of λ g 220
nm, the absorptions listed above for the early intermediates
decreased, and several new weak product bands were observed.
2
When the same experiment was conducted with O as the matrix
material, the same new photochemical product absorptions were
observed and with increased intensity (Figure S3, Supporting
Information). These results indicated that two different processes
were occurring. First was the decomposition of the early
intermediates, and second was the photodetachment of an O
atom from ozone upon irradiation followed by O atom reaction
with an available substrate molecule. The reaction of O atoms
with alkenes has been proposed to occur as shown in Figure 5,
with O atom attack occurring at the less substituted carbon atom
in the double bond.28 This can lead to either stabilization and
formation of the appropriate ketone (3,3-dimethyl-2-butanone
in the system under study here) or the formation of a three-
membered epoxide ring.
-1
as well as a broad absorption at 3497 cm , suggesting the
presence of an O-H-containing species. Product identification
can be made from the known infrared spectra in Ar matrixes
for methyl glyoxal, hydroxyacetone, acetone, tetramethyloxirane,
22,23,29,32
and 3,3-dimethyl-2-butanone.
While mostly stable prod-
ucts were formed, these are consistent with the Criegee
mechanism and secondary reactions of the CI. The observation
of tetramethyloxirane and 3,3-dimethyl-2-butanone in this
system may be the result of the loss of an O atom from the CI
and subsequent reaction with the alkene. Other final products
are in agreement with a related gas-phase study of the reaction
of DMB with ozone at room temperature in a static system with
rapid mixing that yielded methyl glyoxal, methyl acetate, and
hydroxyacetone as major products. Niki et al. noted that the
Using O
2
as the matrix material is thought to increase the
mobility of the O atoms and, as a result, increase the likelihood

 Coleman, Bridgett E.
Coleman, Bridgett E.
 Ault, Bruce S.
Ault, Bruce S.