The Journal of Organic Chemistry
Note
ASSOCIATED CONTENT
Supporting Information
■
*
S
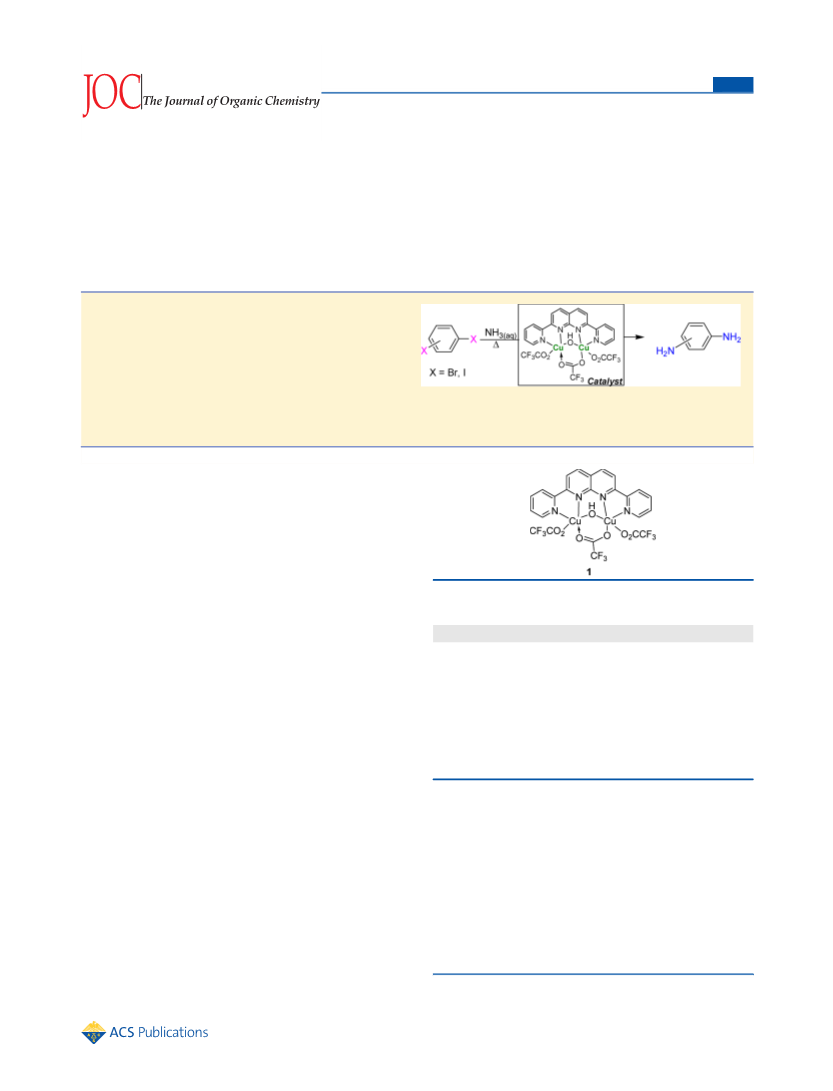
1
AUTHOR INFORMATION
■
*
Notes
The authors declare no competing financial interest.
ACKNOWLEDGMENTS
We thank the National Science Council for financial support
NSC100-2113-M-002-001-MY3).
■
(
Figure 1. Product distribution during the amination of o-C H Br .
6
4
2
REFERENCES
■
(
1) Lawrence, S. A. Amines: Synthesis Properties and Applications;
Cambridge University Press: Cambridge, 2004; pp 104−109.
(2) Recent reviews: (a) Hartwig, J. F. Acc. Chem. Res. 2008, 41,
1534−1544. (b) Surry, D. S.; Buchwald, S. L. Angew. Chem., Int. Ed.
2008, 47, 6338−6361.
1
3
,4-Diaminoanisole (ref 9): 34.2 mg, 99%; H NMR (400 MHz,
CDCl ) δ 6.62 (d, J = 8.4 Hz, 2 H), 6.30 (s, 1 H), 6.24 (d, J = 8.4 Hz,
3
13
1
1
H), 3.71 (s, 3 H), 3.20 (br, 4 H); C NMR (100 MHz, CDCl ) δ
40.3, 140.1, 136.5, 111.9, 104.6, 103.1, 56.3; ESI-MS m/z M 138.16
3
+
(3) Recent reviews: (a) Ma, D.; Cai, Q. Acc. Chem. Res. 2008, 41,
1450−1460. (b) Monnier, F.; Taillefer, M. Angew. Chem., Int. Ed.
(
calcd for C H N O 138.08).
7 10 2
1
2
008, 47, 3096−3099. (c) Evano, G.; Blanchard, N.; Toumi, M. Chem.
2
,4-Diaminoanisole (ref 10): 27.3 mg, 79%; H NMR (400 MHz,
Rev. 2008, 108, 3054−3131. (d) Monnier, F.; Taillefer, M. Angew.
Chem., Int. Ed. 2009, 48, 6954−6971.
CDCl ) δ 6.50 (d, J = 9.2 Hz, 2 H), 6.12 (s, 1 H), 6.00 (d, J = 9.2 Hz,
3
13
1
1
H), 3.90 (br, 4 H), 3.79 (s, 3H); C NMR (100 MHz, CDCl ) δ
40.7, 140.4, 136.9, 112.1, 104.6, 103.3, 56.3; ESI-MS m/z M 138.07
3
+
(4) (a) Xu, H.-J.; Liang, Y.-F.; Cai, Z.-Y.; Qi, H.-X.; Yang, C.-Y.;
Feng, Y.-S. J. Org. Chem. 2011, 76, 2296−2300. (b) Jiao, J.; Zhang, X.-
R.; Chang, N.-H.; Wang, J.; Wei, J.-F.; Shi, X.-Y.; Chen, Z.-G. J. Org.
Chem. 2011, 76, 1180−1183. (c) Chen, J.; Yuan, T.; Hao, W.; Cai, M.
Tetrahedron Lett. 2011, 52, 3710−3713. (d) Zhu, Y.; Wei, Y. Can. J.
Chem. 2011, 89, 645−649. (e) Elmkaddem, M. K.; Fischmeister, C.;
Thomas, C. M.; Renaud, J. L. Chem. Commun. 2010, 46, 925−927.
(
calcd for C H N O 138.08).
7 10 2
1
2
,5-Dimethyl-1,4-benzenediamine (ref 11): 34.1 mg, 100%; H
NMR (400 MHz, CDCl ) δ 6.45 (s, 2 H), 3.5 (br, 4 H), 2.09 (s, 6 H);
3
1
3
C NMR (100 MHz, CDCl ) δ 136.2, 121.4, 117.6, 17.5; ESI-MS m/z
3
+
M 136.13 (calcd for C H N 136.10).
8
12
2
,6-Dimethyl-1,3-benzenediamine (ref 12): 33.7 mg, 99%; 1H
4
(
f) Wu, Z.; Jiang, Z.; Wu, D.; Xiang, H.; Zhou, X. Eur. J. Org. Chem.
NMR (400 MHz, CDCl ) δ 6.7 (s, 1 H), 6.06 (s, 1 H), 3.4 (br, 4 H),
3
.05 (s, 6 H); 13C NMR (100 MHz, CDCl ) δ 142.6, 132.9, 112.3,
2010, 1854−1857. (g) Lundgren, R. J.; Peters, B. D.; Alsabeh, P. G.;
Stradiotto, M. Angew. Chem., Int. Ed. 2010, 49, 4071−4074. (h) Xu,
H.; Wolf, C. Chem. Commun. 2009, 3035−3037. (i) Wang, D. P.; Cai,
Q.; Ding, K. Adv. Synth. Catal. 2009, 351, 1722−1726. (j) Jiang, L.; Lu,
X.; Zhang, H.; Jiang, Y.; Ma, D. J. Org. Chem. 2009, 74, 4542−4546.
2
1
3
+
02.5, 16.3; ESI-MS m/z M 136.21 (calcd for C H N 136.10).
8
12
2
3
-Chloroaniline: identical to the commercial available sample (17.6
1
mg, 55%); H NMR (400 MHz, CDCl ) δ 6.98−7.01 (m, 1 H), 6.71
d, J = 7.6 Hz, 1 H), 6.63 (s, 1 H), 6.54 (d, J = 8 Hz, 1 H), 3.85 (br s, 2
H); C NMR (100 MHz, CDCl ) δ 148.0, 134.8, 130.4, 118.4, 115.1,
3
(
(
k) Yang, C. T.; Fu, Y.; Huang, Y. B.; Yi, J.; Guo, Q. X.; Liu, L. Angew.
13
3
Chem., Int. Ed. 2009, 48, 7398−7401. (l) Wu, X. F.; Darcel, C. Eur. J.
1
13.1.
Org. Chem. 2009, 4753−4756. (m) Schulz, T.; Torborg, C.; Enthaler,
1
2
,6-Diaminopyridine (ref 13): 26.2 mg, 96%; H NMR (400 MHz,
̈
S.; Schaffner, B.; Dumrath, A.; Spannenberg, A.; Neumann, H.; Bcrner,
CDCl ) δ 7.23 (t, J = 7.8 Hz, 1 H), 5.89 (d, J = 7.8 Hz, 2 H), 4.20 (br
3
A.; Beller, M. Chem.Eur. J. 2009, 15, 4528−4533. (n) Xia, N.;
1
s, 4 H); H NMR (400 MHz, DMSO-d ) δ 7.00 (t, J = 8 Hz, 1 H),
6
Taillefer, M. Angew. Chem., Int. Ed. 2009, 48, 337−339.
13
5
.63 (d, J = 8 Hz, 2 H), 5.40 (br s, 4 H); C NMR (100 MHz,
(
5) (a) Surry, D. S.; Buchwald, S. L. J. Am. Chem. Soc. 2007, 129,
13
CDCl ) δ 156.4, 136.3, 94.0; C NMR (100 MHz, DMSO-d ) δ
1
3
6
10354−10355. (b) Lee, S.; Jørgensen, M.; Hartwig, J. F. Org. Lett.
+
57.8, 137.8, 95.0; ESI-MS m/z M 109.10 (calcd for C H N 109.06).
5 7 3
2001, 3, 2729−2732.
3
,5-Diaminopyridine: identical to the commercial available sample
(
6) Shen, Q.; Hartwig, J. F. J. Am. Chem. Soc. 2006, 128, 10028−
1
(
26.0 mg, 95%); H NMR (400 MHz, CDCl ) δ 7.45 (s, 2 H), 6.35 (s,
H), 3.7 (br s, 4 H); H NMR (400 MHz, acetone-d ) δ 7.35 (s, 2 H),
6
3
10029.
1
1
6
1
(7) Liao, B.-S.; Liu, Y.-H.; Peng, S.-M.; Liu, S.-T. Dalton Trans. 2012,
41, 1158−1164.
13
.30 (s, 1H), 4.49 (br, 4 H); C NMR (100 MHz, CDCl ) δ 142.9,
3
27.9, 107.3; 13C NMR (100 MHz, acetone-d ) δ 144.8, 126.7, 105.9;
6
(8) Touil, M.; Elhabiri, M.; Lachkar, M.; Siri, O. Eur. J. Org. Chem.
2011, 1914−1921.
+
ESI-MS m/z M 109.17 (calcd for C H N 109.06).
5
7
3
1
Biphenyl-4,4′-diamine (ref 4a): 46.0 mg, 100%; H NMR (400
(9) Keurulainen, L.; Salin, O.; Siiskonen, A.; Kern, J. M.; Alvesalo, J.;
Kiuru, P.; Maass, M.; Yli-Kauhaluoma, J.; Vuorela, P. J. Med. Chem.
2010, 53, 7664−7674.
MHz, CDCl ) δ 7.33 (d, J = 8.8 Hz, 4H), 6.71 (J = 8.8 Hz, 4H), 3.4
3
(
br, 4H); 13C NMR (100 MHz, CDCl ) δ 144.4, 131.4, 126.9, 115.1;
3
+
ESI-MS m/z M 184.12 (calcd for C H N 184.10).
(10) Rahaim, R. J., Jr.; Maleczka, R. E., Jr. Synthesis 2006, 3316−
3340.
1
2
12
2
1
,6-Diamino-2-methoxynaphthalene: 47 mg, 100%; 1H NMR
400 MHz, CDCl ) δ 7.60 (d, J = 10 Hz, 1 H), 7.12 (d, J = 8.8 Hz, 1
(
(11) Eilstein, J.; Arnau, E. G.; Duche, D.; Rousset, F.; Lepoittevin, J.
P. Chem. Res. Toxicol. 2006, 19, 1248−1256.
(12) Mousseau, J. J.; Xing, L.; Tang, N.; Cuccia, L. A. Chem.Eur. J.
2009, 15, 10030−10038.
(13) Elmkaddem, M. K.; Fischmeister, C.; Thomas, C. E. M.;
Renaud, J.-L. Chem. Commun. 2010, 46, 925−927.
(14) Jeong, M. J.; Park, J. H.; Lee, C.; Chang, J. Y. Org. Lett. 2006, 8,
2221−2224.
3
H), 7.05 (d, J = 8.8 Hz, 1 H), 6.80−6.92 (m, 2H), 3.90 (s, 3 H), 3.8
(
br, 4 H); 13C NMR (100 MHz, CDCl ) δ 142.1, 130.7, 129.8, 121.6,
3
1
21.5, 118.5, 117.6, 116.1, 114.6, 109.3, 57.5; ESI-HRMS (TOF) m/z
+
M 188.0947 (calcd for C H N O 188.0950).
1
1
12
2
1
1
,3,5-Benzenetriamine (ref 14): 30.7 mg, 100%; H NMR (400 Hz,
13
CDCl ) δ 5.52 (s, 3 H), 1.50 (br, 6 H); C NMR (100 MHz, CDCl3)
δ 148.2, 93.6; ESI-MS m/z M 123.21 (calcd for C H N 123.08).
3
+
6
9
3
D
dx.doi.org/10.1021/jo301244p | J. Org. Chem. XXXX, XXX, XXX−XXX

 Liao, Bei-Sih
Liao, Bei-Sih
 Liu, Shiuh-Tzung
Liu, Shiuh-Tzung