10.1002/cssc.201900814
ChemSusChem
COMMUNICATION
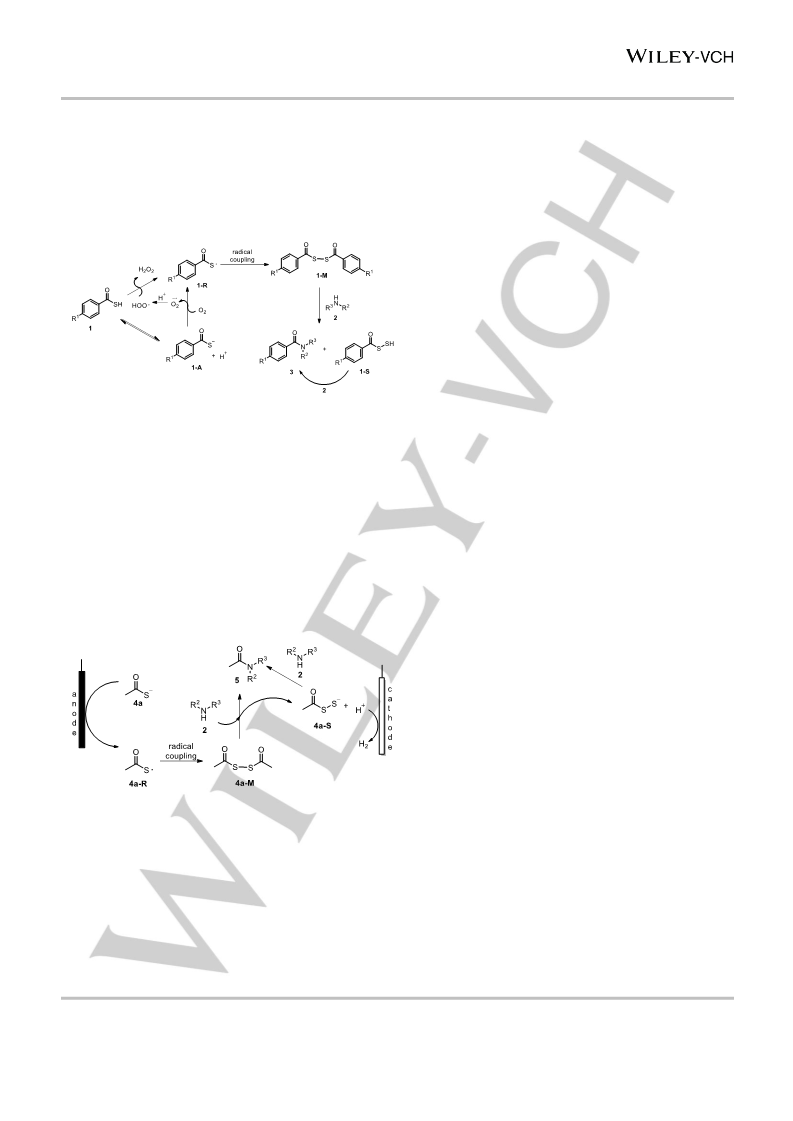
produces another benzoylthio radical 1-R. Next, the self-coupling
of 1-R forms the disulfide 1-M. The nucleophilic
addition/elimination of amine 2 with 1-M to give the desired
product amide 3 and the intermediate 1-S. 1-S can be further
subjected to aminolysis to give 3, which can be supported by the
experimental results, since the reaction of equal equivalent of 1a
and 2a gave amide 3aa in a yield of 63% (SI, Table S1, entry 1).
thioaliphatic acids by spontaneous reaction and electrosynthesis,
respectively.
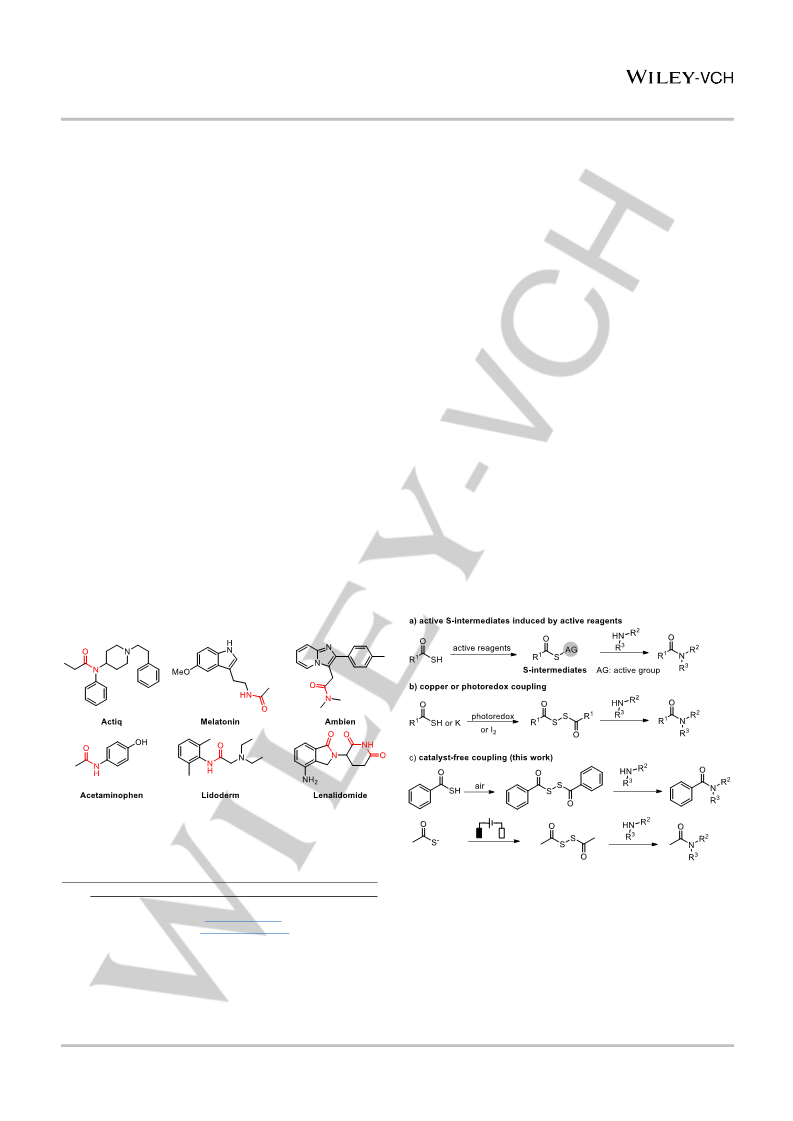
In summary, we have developed a mild catalyst-free amide
bond forming strategy by using various thiocarboxylic acids as the
acyl source. The reaction is proposed to proceed through a
disulfide intermediate that reacts with an amine to provide an
amide. It is noteworthy that in the presence of air, the reaction of
thiobenzoic acids with various amines can occur spontaneously,
while the reaction of thioaliphatic acids with amines can be easily
carried out by electrosynthesis. By this method, various amides
can be readily synthesized in excellent yields without using any
catalyst and activator. The successful synthesis of bioactive
compounds also highlights the synthetic utility of this strategy in
medicinal chemistry.
Acknowledgements
This work was financially supported by the National Natural
Science Foundation of China (Nos 21472152 and 21672174), the
Basic and Frontier Research Project of Chongqing
(cstc2015jcyjBX0106) and the Innovation Foundation of
Chongqing City for Postgraduate (CYB18097).
Scheme 7. A possible mechanism for the spontaneous reaction.
For the process of forming amides by electrosynthesis
(Scheme 8), potassium thioacetate 4a (Eox = +0.33 V vs SCE in
MeCN) (SI, Figure S1) is electron-oxidized to the acetylthio
radical 4a-R by anode. The radical coupling of 4a-R forms the
disulfide 4a-M, which reacts with amine 2 (Eox = +1.15 V vs SCE
in MeCN) (SI, Figure S1) to afford acetyl amide 5 and
intermediate 4a-S. The experimental fact that the reaction of equal
equivalent of 4a and 2a gave amide 5aa in a yield of 66% (SI,
Table S5) indicted that the desired product 5 can also be
produced by the aminolysis of 4a-S12. The proton from aminolysis
can be reduced by cathode to form H2.
Keywords: catalyst free • electrosynthesis • amide bond •
thiocarboxylic acids • disulfide
[1]
a ) C. A. G. N. Montalbetti, V. Falque, Tetrahedron 2005, 61, 10827-
10852; b ) E. Valeur, M. Bradley, Chem. Soc. Rev. 2009, 38, 606-631;
c ) C. L. Allen, J. M. Williams, Chem. Soc. Rev. 2011, 40, 3405-3415; d )
V. R. Pattabiraman, J. W. Bode, Nature 2011, 480, 471-479; e ) R. M. de
Figueiredo, J. S. Suppo, J. M. Campagne, Chem. Rev. 2016, 116,
12029-12122; f ) A. Ojeda-Porras, D. Gamba-Sanchez, J. Org. Chem.
2016, 81, 11548-11555; g ) L. Tang, X. M. Li, J. H. Matuska, Y. H. He, Z.
Guan, Org. Lett. 2018, 20, 5618-5621.
[2]
J. S. Carey, D. Laffan, C. Thomson, M. T. Williams, Org. Biomol. Chem.
2006, 4, 2337-2347.
[3]
[4]
L. M. Jarvis, Chem Eng News 2018, 96, 26-30.
N. N, V. M. Thimmalapura, B. Hosamani, G. Prabhu, L. R. Kumar, V. V.
Sureshbabu, Org. Biomol. Chem. 2018, 16, 3524-3552.
[5]
[6]
D. Crich, I. Sharma, Angew. Chem., Int. Ed. 2009, 48, 2355-2358.
a ) Y. Rao, X. Li, S. J. Danishefsky, J. Am. Chem. Soc. 2009, 131, 12924-
12926; b ) X. Wu, J. L. Stockdill, P. K. Park, S. J. Danishefsky, J. Am.
Chem. Soc. 2012, 134, 2378-2384.
[7]
a ) F. B. Dyer, C. M. Park, R. Joseph, P. Garner, J. Am. Chem. Soc. 2011,
133, 20033-20035; b ) S. M. Mali, S. V. Jadhav, H. N. Gopi, Chem
Commun (Camb) 2012, 48, 7085-7087; c ) S. M. Mali, R. D. Bhaisare, H.
N. Gopi, J. Org. Chem. 2013, 78, 5550-5555.
Scheme 8. A possible mechanism for the electrosynthesis.
[8]
W. Chen, J. Shao, M. Hu, W. Yu, M. A. Giulianotti, R. A. Houghten, Y.
Yu, Chem. Sci. 2013, 4, 970-976.
In the mechanisms proposed above, the formation of acylthio [9]
radicals is a critical step. Structurally, the benzoylthio radical 1-R
(Scheme 7) is more stable than acetylthio radical 4a-R (Scheme
8), which means that 1-R is easier to form than 4a-R. Thus, we
speculate that under our reaction conditions, 1-R can be easily
produced by auto-oxidation of thiobenzoic acids 1, while the
formation of 4a-R in high yields requires electrooxidation of
thioacetate 4a. This speculation can explain the experimental
J. Pan, N. O. Devarie-Baez, M. Xian, Org. Lett. 2011, 13, 1092-1094.
[10] M. K. Miraki, E. Yazdani, L. Ghandi, K. Azizi, A. Heydari, Appl.
Organomet. Chem. 2017, 31, e3744.
[11] S. M. Mali, H. N. Gopi, J. Org. Chem. 2014, 79, 2377-2383.
[12] H. X. Liu, L. Y. Zhao, Y. F. Yuan, Z. F. Xu, K. Chen, S. X. Qiu, H. B. Tan,
Acs. Catal. 2016, 6, 1732-1736.
[13] S. Das, S. Ray, A. B. Ghosh, P. K. Samanta, S. Samanta, B. Adhikary,
P. Biswas, Appl. Organomet. Chem. 2018, 32, e4199.
[14] R. A. Sheldon, Green Chem. 2007, 9, 1273-1283.
results of synthesizing amide from thiobenzoic acids and [15] a ) S. Mohle, M. Zirbes, E. Rodrigo, T. Gieshoff, A. Wiebe, S. R.
Waldvogel, Angew. Chem., Int. Ed. 2018, 57, 6018-6041; b ) A. Wiebe,
This article is protected by copyright. All rights reserved.

 Tang, Li
Tang, Li
 Matuska, Jack H.
Matuska, Jack H.
 Huang, Yu-Han
Huang, Yu-Han
 He, Yan-Hong
He, Yan-Hong
 Guan, Zhi
Guan, Zhi