390
M. Mokhtary et al. / C. R. Chimie 15 (2012) 389–393
PVPP-BF3
added PVPP-BF3 (0.05 g) and stirring continued at room
temperature. The progress of the reaction was monitored
by TLC. After completion of the reaction, dichloromethane
(15 ml) was added and the reaction mixture was filtered.
The organic phase was washed with saturated NaHCO3
solution (15 ml), dried over anhydrous Na2SO4 and
evaporated to give the pure acylated product. The isolated
products were characterized by spectral studies (FT-IR and
NMR). The spectra were compared with those of standard
esters. The reaction times and yields of the products are
presented in Table 3.
R
OR' + Ac2O
R= alkyl, benzyl, aryl
R'= H, SiMe3
R
OAc
CH3CN/ r.t.
Scheme 1. Acylation of alcohols, phenols and trimethylsilyl ethers in
acetonitrile via PVPP-BF3.
of our research on amidation of benzhydrol with nitriles via
polyvinylpolypyrrolidone-bound boron trifluoride (PVPP-
BF3)[25], inthisworkwefoundthatPVPP-BF3 can beusedas
a recyclable and more efficient catalyst for acetylation of
alcohols, phenols and trimethylsilyl ethers in excellent
yields at room temperature (Scheme 1).
2.4. Selected characterization data
2. Experimental
2.4.1. 2-phenylethyl acetate (Table 3, entry 1)
Yield 99%; IR (KBr)
1375, 1037, 746 cmÀ1. 1H NMR (CDCl3)
(t, 2H, J = 7.2), 4.29 (t, 2H, J = 7.2), 7.24–7.35 (m, 5H) ppm.
13C NMR (CDCl3)
= 170.96, 137.80, 128.85, 128.47,
y
: 2958, 2925, 1745, 1529, 1457,
All chemicals were purchased from Merck chemical
company. Melting points were recorded on an electro
thermal melting point apparatus. The NMR spectra were
recorded in CDCl3 with TMS as an internal standard on a
Bruker advance DRX 400 MHz spectrometer. IR spectra
were determined on a SP-1100, P-UV-Com instrument.
Purity determination of the products was accomplished by
TLC on silica gel poly gram SIL G/UV 254 plates. Products
were identified by comparison IR and NMR spectra, with
those reported for authentic samples.
d
= 2.06 (s, 3H), 2.95
d
126.53, 64.89, 34.07, 20.93.
2.4.2. 4-isopropylbenzyl acetate (Table 3, entry 4)
Yield 97%; IR (KBr)
1027, 819, 736 cmÀ1. 1H NMR (CDCl3)
(s, 3H), 2.9 (m, 1H), 4.68 (s, 2H), 7.24 (d, 2H, J = 8.4), 7.31 (d,
2H, J = 8.4) ppm. 13C NMR (CDCl3)
= 171.02, 151.24,
y: 2958, 2861, 1743, 1457, 1230,
d
= 1.27 (d, 6H), 2.12
d
136.10, 127.32, 126.59, 72.47, 34.55, 24.85, 20.11.
2.1. Catalyst preparation
2.4.3. p-nitrobenzene acetate (Table 3, entry 14)
In this method, boron trifluoride etherate was immo-
Yield 99%; IR (KBr)
1197, 1099, 858 cmÀ1. 1H NMR (CDCl3)
(d, 2H, J = 7.2), 8.27 (d, 2H, J = 7.2) ppm. 13C NMR (CDCl3)
= 168.46, 155.36, 146.15, 125.24, 122.48, 21.16.
y
: 2927, 2852, 1762, 1523, 1344,
bilized on polyvinylpolypyrrolidone to give
a
stable
d
= 2.37 (s, 3H), 7.28
polymeric Lewis acid reagent according to our previous
article [25]. To a suspension of PVPP (3 g) in CH2Cl2 (25 ml),
a solution of BF3.OEt2 (5 ml) in CH2Cl2 (15 ml) was added
dropwise and the mixture stirred for 1 h at room
temperature. The resulting resin was filtered and washed
with CH2Cl2 (2 Â 10 ml) and dried in a vacuum desiccator
to give a stable and non-hygroscopic powder.
d
2.4.4. m-tolyl acetate (Table 3, entry 15)
Yield 99%; IR (KBr) : 2927, 2861, 1770, 1589, 1457,
1371, 1145, 1014, 943, 784 cmÀ1. 1H NMR (CDCl3)
= 2.31
(s, 3H), 2.38 (s, 3H), 6.9-7.3 (m, 3H) ppm. 13C NMR (CDCl3)
= 169.85, 149.42, 135.53, 129.99, 125.18, 121.28, 118.63,
y
d
d
2.2. General procedure for acetylation of alcohols and phenols
21.16, 20.91.
To a stirred mixture of the alcohol (1 mmol), Ac2O
(2 mmol) and CH3CN (3 ml) was added PVPP-BF3 (0.025 g)
and stirring continued at room temperature. The progress
of the reaction was monitored by TLC. After completion of
the reaction, dichloromethane (15 ml) was added and the
reaction mixture was filtered. The organic phase was
washed with saturated NaHCO3 solution (15 ml), dried
over anhydrous Na2SO4 and evaporated to give the pure
acylated product. The isolated products were character-
ized by spectral studies (FT-IR and NMR). The spectra were
compared with those of standard esters. The yield was
calculated as mmol of purified product with respect to
mmol of initial alcohol. The reaction times and yields of the
products are presented in Table 3.
2.3. General procedure for acetylation of silylated alcohols
and phenols
To a stirred mixture of the silylated alcohol or silylated
phenol (1 mmol), Ac2O (2 mmol) and CH3CN (3 ml) was
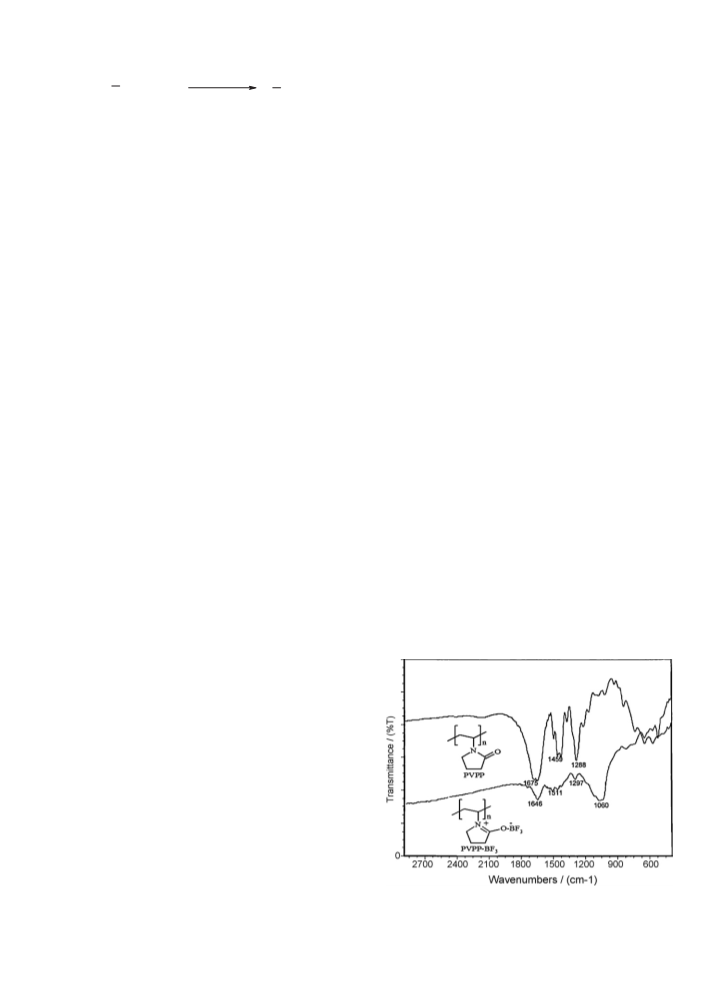
Fig. 1. The FT-IR spectrum of polyvinylpolypyrrolidone (PVPP) and
(PVPP-BF3) complex.

 Mokhtary, Masoud
Mokhtary, Masoud
 Qandalee, Mohammad
Qandalee, Mohammad
 Najafizadeh, Faranak
Najafizadeh, Faranak