Carbon Supported Pd Nanocrystals as High Efficient Catalyst
1323
50 mg of the sample was reduced in a 10 % H2/Ar gas
mixture at 523 K (with the heating rate of 10 K/min) for 1 h
under a flow rate of 50 mL/min. Once the catalysts were
cooled down to room temperature, the surface was purged in
an argon flow for 1 h. Then, the TPD were conducted in
flowing Ar at 10 K/min to 825 K.
reactor. The autoclave was sealed and purged with N2 at
least five times to expel air and then H2 was introduced into
the reactor to replace N2. The reaction was started after the
reaction temperature increased to the set point and the H2
pressure was modified to the desired value.
The reaction process was monitored by taking small
amounts of the reaction mixture at set time intervals. The
samples were analyzed by GC (Agilent 6890, FID detector)
with a 30 m HP-5 capillary column and PEG-20 M capil-
lary column using a temperature ramp from 423 to 483 K
at rate of 5 K/min. The calibrated area normalization
method was used. The separations of different products
were performed by using column chromatography on silica
gel (n-hexane/EtOAc, 50:1) and the structural characteris-
tics of products were accomplished by GC–MS (GCT
CA156) and 1H NMR (Varian INOVA). Details are
described in the electronic supporting information (ESI).
2.2.5 Elemental Analysis (ICP-AES and Energy Dispersive
Spectroscopy (EDS))
The real percentage of Pd loading on different supports was
detected by ICP-AES (Varian VISTA- MPX). Briefly,
0.1 g of each samples were dissolved in aqua regia and
diluted into 1 mg/ml solution and then performed the ICP
detection.
The amounts of chlorine anion on the surface of Pd/
activated carbon were detected by the EDS which was
recorded with a JEOL 7500F field emission scanning
electron microscope. EDS line scan data were collected
using an acceleration voltage of 200 kV with a 0.5 nm
diameter probe. Integrated intensities obtained from the Pd
L and Cl K lines were used for quantification of the two
elements, respectively.
3 Results and Discussion
3.1 Comparison of Different Catalytic Systems
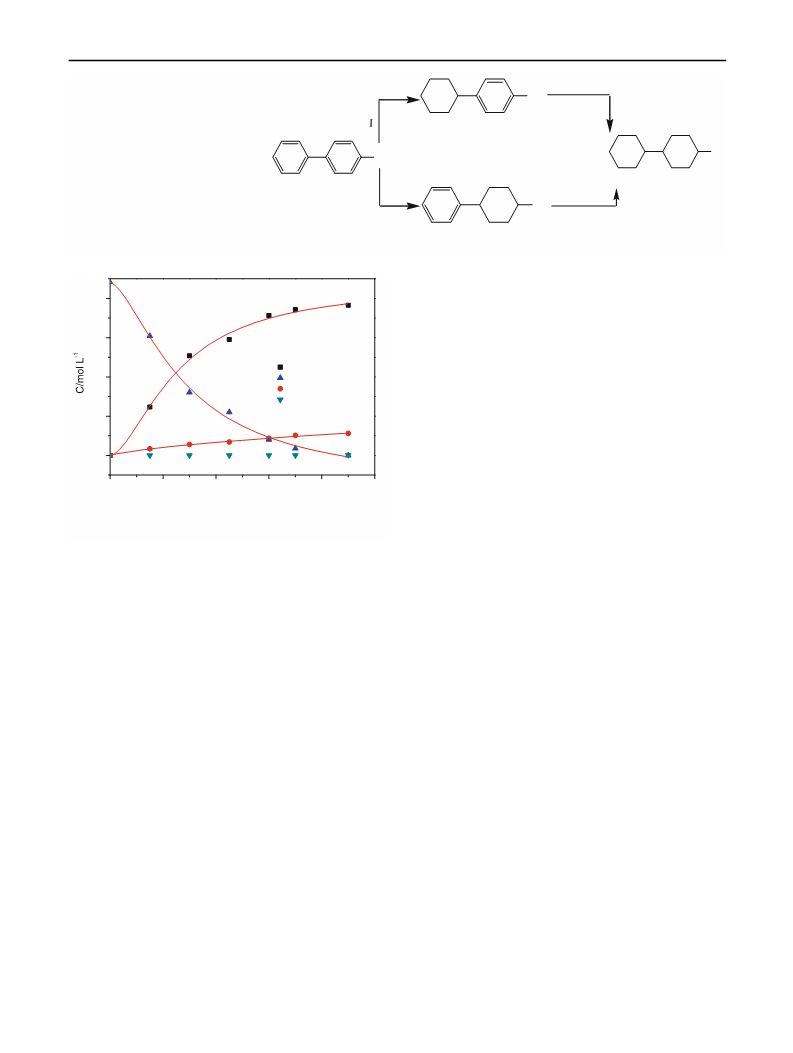
The performance of six catalytic systems in the hydroge-
nation of p-PP is summarized in Table 1. Obviously, all the
Pd-based catalysts exhibited higher selectivity to p-CP
(over 75 %) while 5 wt% Pt/AC and 5 wt% Ru/AC pre-
ferred the deep hydrogenation of p-PP and Skeletal Ni
slightly inclined to produce p-PC. The trans-/cis- ratios of
p-PC and p-CC are dominated by the kind of metal used in
the catalyst and, generally, the cis-configuration of p-PC
and trans-configuration of p-CC are favored. Further
compared the three kinds of 5 wt% Pd/AC, though the
commercial 5 wt% Pd/AC (supported by Tongyong
Chemicals Dalian, China) and the 5 wt% Pd/AC prepared
according to the method mentioned in ref [12] resulted in
acceptable selectivity to p-CP, however, the activity were
much lower than the 5 wt% Pd/AC prepared in this paper.
Therefore, take both the activity and selectivity into con-
sideration; the Pd/AC catalyst prepared this time is the
suitable catalyst for selective hydrogenation of p-PP to
produce p-CP.
2.2.6 Boehm Titration
The quantity of the surface oxygen-containing groups for
carbon supports were measured according to the method
suggested by Boehm [15–17]. Generally, 0.25 g of each
kind of carbon support was placed in a bottle, to which
25 ml of 0.1 N solution of Na2CO3, NaHCO3 or NaOH
was added, and the bottles were sealed. After shaking for
24 h, 20 ml of the solution were back-titrated using
0.05 NÁHCl. The amounts of NaOH and NaHCO3 reacted
were used to calculate the total number of acidic groups
(phenol, lactone, and carboxylic groups) and the number of
carboxylic groups, respectively. Na2CO3 consumption
indicated the amount of both lactone and carboxylic
groups. Consequently, the number of lactone groups was
determined from the difference in Na2CO3 and NaHCO3
consumption. The number of phenol groups was deter-
mined from the difference between NaOH and Na2CO3
consumption. The overall quantity of basic groups was
determined by titration with 0.1 N HCl and back-titrated
with 0.05 N NaOH.
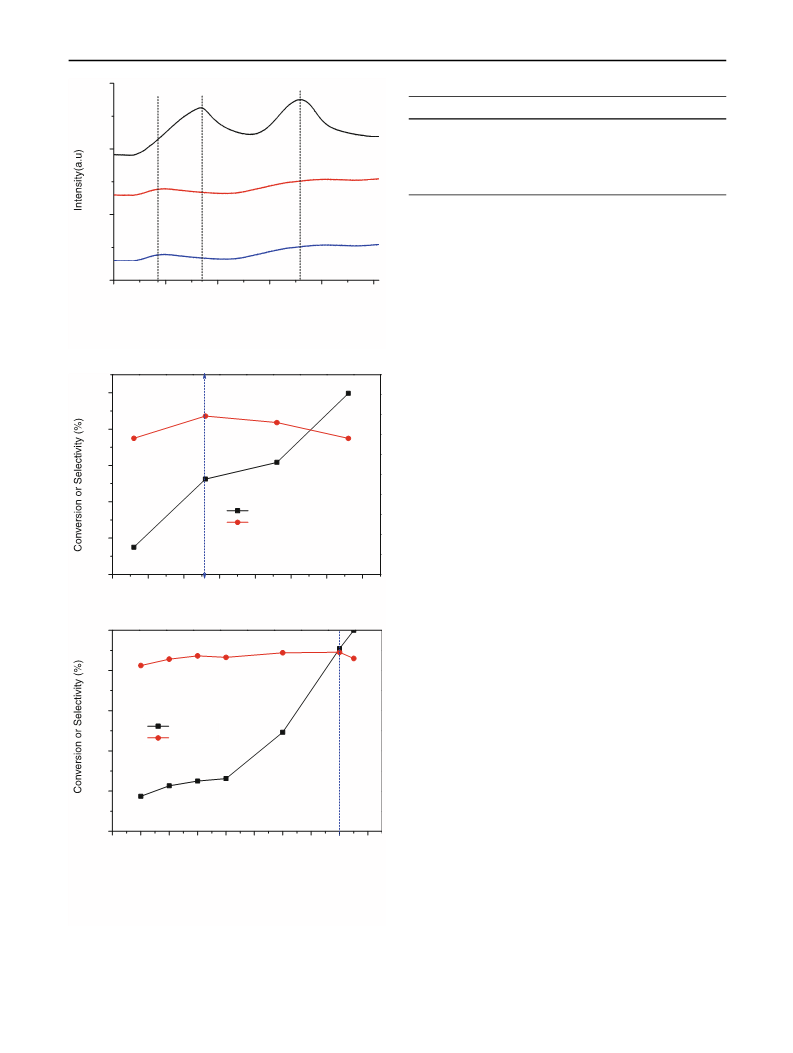
3.2 Effects of Solvents
The effects of different solvents are listed in Table 2. As
shown, the selectivity to p-CP was over 70 % when using
polar solvents as the reaction medium and the highest
selectivity was obtained with THF. In contrast, when using
cyclohexane as solvent, the selectivity to p-CP was only
12 % even though the highest activity was obtained in this
condition. These results maybe caused by the different
properties of solvents and the structural characteristic of
2.3 Hydrogenation Reaction
The hydrogenation reaction was carried out in a 70 mL
stainless steel autoclave equipped with a magnetic stirrer.
Catalysts were washed with deionized water and solvent
five times before being added to the reactor. Then the p-PP,
solvent and catalyst were quantitatively introduced to the
123

 Liu, Chengyun
Liu, Chengyun
 Lu, Lianhai
Lu, Lianhai
 Rong, Zeming
Rong, Zeming
 Liang, Changhai
Liang, Changhai
 Wang, Yue
Wang, Yue
 Qu, Qingping
Qu, Qingping