7108
A.S. Ivanov et al. / Tetrahedron 65 (2009) 7105–7108
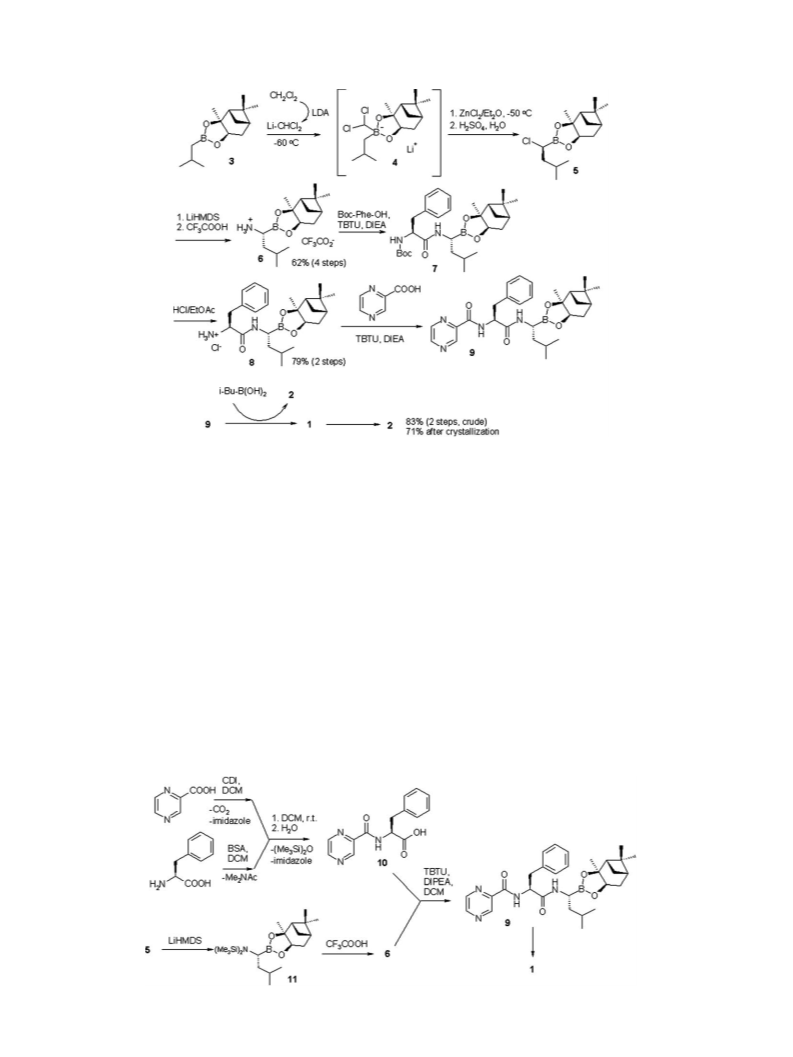
3
1
.3. (1S,2S,3R,5S)-Pinanediol 1-ammonium-3-methylbutane-
-boronate trifluoroacetate (8)
NH), 7.10–7.33 (m, 5H, CHPh), 4.93 (m, 1H,
8.6,1.8 Hz, CHpinane–O), 3.2 (t, 2H, CH Ph), 3.05–0.7 (m, 25H,
B, 5CH , 3CH , 3CH); (75 MHz, CDCl ) 170.5, 162.5, 147.1, 144.0,
142.5, 136.3, 129.2 (2C), 128.2 (2C), 126.6, 85.3, 77.5, 53.7, 51.3, 39.8
a
CHPhe), 4.30 (dd, 1H, J
2
a
CH–
3
2
d
C
3
Bis(trimethylsilyl)amine 11 (10.00 g, 62 mmol) was dissolved in
diethyl ether (90 mL), and the resulting solution was added during
(CH
(CH
2
), 39.4, 38.5 (CH
), 25.9, 23.8, 22.7, 21.8.
2 2
), 38.3, 37.9, 35.7, 35.4 (CH ), 28.4, 26.9, 26.1
1
h to a stirred solution of trifluoroacetic acid (7.5 mL, 98 mmol) in
2
ꢀ
diethyl ether (100 mL) at ꢁ15 to ꢁ10 C under argon. The mixture
was stirred for another 1.5 h, and the precipitate formed was filtered,
ꢀ
3.6. Bortezomib (2)
washed with diethyl ether (50 mL) and dried under vacuum at 35 C
to give 7.2 g (78%) of the white crystalline salt 8, mp 197–198 C. The
ꢀ
Boronic ester 9 (68 g, 13.12 mmol) was dissolved in methyl al-
cohol (0.5 L). To that solution hexane (0.5 L) and 1 N hydrochloric
acid (290 mL) were added under stirring, and the biphasic mixture
product was stored in the fridge under argon until used on the next
step.
ꢀ
was cooled to 10 C. Isobutylboronic acid (24 g, 235 mmol) was
3
.4. N-Pyrazinoyl-L-phenylalanine (10)
added, and the mixture was stirred for 17 h at room temperature.
The layers were separated in the separating funnel. The methanolic
layer was washed with hexane (3ꢂ0.1 L) and the combined hexane
solutions were evaporated to dryness. From the residual oil boronic
ester 3, incorporating valuable (1S,2S,3R,5S)-pinanediol chiral aux-
hiliary, was regenerated by chromatography. The aqueous meth-
anolic layer was neutralized with sodium hydrocarbonate (30 g)
and extracted with ethyl acetate (0.85 L). The solution was dried
over sodium sulfate, filtered through a plug of silica gel, and
recrystallized twice from ethyl acetate to give 26.55 g (26.3 mmol,
N,O-Bis(trimethylsilyl)-
by adding BSA (50.7 g, 250 mmol) to a suspension of
L
-phenylalanine solution was perepared
L-phenylala-
nine (20.5 g, 125 mmol) in DCM (200 mL), and stirring the resulting
solution at room temperature overnight.
A solution of pyrazinecarboxylic acid imidazolide was obtained
by adding CDI (26.5 g, 250 mmol) to a stirred suspension of pyr-
azinecarboxylic acid (24.0 g, 194 mmol) in DCM (400 mL). The
mixture was stirred at room temperature overnight, cooled to ꢁ30
ꢀ
to ꢁ40 C, and then N,O-bis(trimethylsilyl)-(
L)-phenylalanine so-
6
0%) of the white amorphous solid 99.83% pure by HPLC (UV).
(KBr): 701, 750, 1021, 1155, 1201, 1400, 1522, 1657, 2954, 3385;
360 MHz, CDCl ) 9.08 (d, 1H, J 1.2 Hz), 8.82 (d, 2H, J 2.4 Hz), 8.80 (s,
H), 8.70 (m, 1H), 7.10–7.30 (m, 5H, CHPh), 4.86 (m, 1H, CHPhe),
.15 (m, 2H, CH Ph), 2.64 (br m, 1H, CH–B), 1.52 (m, 1H, CH(CH ),
– Pr), 0.76 (d, 6H, J 6.60 Hz, 2CH ); (90 MHz,
) 172.5, 162.3, 147.5, 143.9, 143.3, 143.2, 143.1, 136.6, 129.1,
n
max
lution was added dropwise during 30 min. The temperature was
ꢀ
d
H
raised to 20 C during 2 h and the mixture was stirred for another
(
1
3
3
1
7 h. The reaction solution was washed with aqueous solution of
citric acid (60 g of citric acid monohydrate in 400 mL of water), the
aqueous phase was separated, washed with methylene dichloride
100 mL), and the combined organic solutions were diluted with
diethyl ether (200 mL), and dried over sodium sulfate. The solvent
a
2
a
3 2
)
i
1.30,1.17 (2m, 2H, CH
2
3
d
C
(
CDCl
3
ꢀ
2 2
127.9, 127.8, 126.2, 51.5, 42.7, 39.8 (CH ), 37.2 (CH ), 24.9, 22.8, 22.3.
was removed under vacuum at 35 C to give 3.1 g (97%) of yellow
ꢀ
powder, mp 142–146 C. [Found: C, 61.85; H, 4.90; N, 15.32.
2
0
C
14
H
13
N
3
O
3
requires C, 61.99; H, 4.83; N, 15.49.] [
max (KBr): 1522, 1653, 1680, 2925, 2952, 3325;
360 MHz, acetone-d ) 9.22 (d, 1H, J 1.4 Hz, CHpyrazine), 8.83 (d,
H, J 2.5 Hz, CHpyrazine), 8.63 (dd, 1H, J 2.5, 1.44 Hz, CHpyrazine),
.39 (br d, 1H, NH), 7.40–7.10 (m, 5H, CHPh), 5.00 (m, 1H, CHPhe),
.34 (m, 2H, CH Ph); (90 MHz, DMSO-d ) 172.4, 162.6, 147.8,
44.1, 143.5, 143.4, 137.4, 129.1 (2C), 128.2 (2C), 126.5, 53.5, 36.3.
a]
D
þ13.5 (c 1.0,
Acknowledgements
MeOH);
n
d
H
(
6
The authors are grateful to Vladimir Korolev, Nikolay Uvarov,
and Ivan Petukhov for chromatographic and spectral analysis.
1
8
3
a
2
d
C
6
1
References and notes
1
. Bross, P. F.; Kane, R.; Farrell, A. T.; Abraham, S.; Benson, K.; Brower, M. E.;
3
.5. (1S,2S,3R,5S)-Pinanediol N-pyrazinoyl-
L-phenylalanine-L-
Bradley, S.; Gobburu, J. V.; Goheer, A.; Lee, S.-L.; Leighton, J.; Liang, C. Y.; Los-
tritto, R. T.; McGuinn, W. D.; Morse, D. E.; Rahman, A.; Rosario, L. A.; Verbois, S.
L.; Williams, G.; Wang, Y.-C.; Pazdur, R. Clin. Cancer Res. 2004, 10, 3954–3964.
. Bonvini, P.; Zorzi, E.; Basso, G.; Rosolen, A. Leukemia 2007, 21, 838–842. doi:10.
boronoleucine (9)
2
3
Ammonium salt 6, (7.1 g, 18.7 mmol), N-pyrazinoyl-L-phenylal-
1038/sj.leu.2404528
anine 10 (5.08 g, 18.7 mmol), and TBTU (6.62 g, 20.6 mmol) were
. Kisselev, A. F.; Goldberg, A. L. Chem. Biol. 2001, 8, 739–758.
ꢀ
suspended in DCM (75 mL), and the mixture was cooled to ꢁ5 C
4. Adams, J. Proteasome Inhibitors in Cancer Therapy; Humana: Totowa, NJ, 2004;
i
pp 17–38.
while stirring. After that a solution of Pr
2
NEt (9.5 mL) in DCM
5. Troy, D. B.; Beringer, P. Remington: The Science and Practice of Pharmacy, 21st ed.;
(
35 mL) was added dropwise during 2 h to a stirred reaction mix-
Lippincott Williams & Wilkins: Philadelphia, PA, 2005.
ꢀ
ture maintaining the temperature between ꢁ10 and ꢁ5 C. The
mixture was stirred for another 1.5 h, and then slowly heated to
room temperature. The solvent was evaporated under vacuum, and
the residue was dissolved in ethyl acetate (110 mL), washed with
water (75 mL), with 3% aqueous potassium carbonate (3ꢂ50 mL),
with water (75 mL), with 3% aqueous citric acid (3ꢂ50 mL), and
lastly, with water (75 mL). The organic phase was dried over so-
dium sulfate, the solvent was removed under vacuum, the residue
dissolved in diethyl ether (50 mL) and filtered through a pad of
silica gel (washed with 100 mL of ether). Ether was evaporated
under vacuum to give 8.14 g (84%) of viscous oil, which slowly
6. Adams, J.; Behnke, M.; Cruickshank, A. A.; Dick, L. R.; Grenier, L.; Klunder, J. M.;
Ma, J.-T.; Plamondon, L.; Stein, R. L. Bioorg. Med. Chem. Lett. 1998, 8, 333–338.
7. Li, Y.; Plesescu, M.; Sheehan, P.; Daniels, J. S.; Prakash, S. R. J. Labelled Compd.
Radiopharm. 2007, 50, 402–406. doi:10.1002/jlcr.1173
8. Pickersgill, I. F.; Bishop, J.; Koellner, C.; Gomez, J.-M.; Geiser, A.; Hett, R.; Am-
moscato, V.; Munk, S.; Lo, Y.; Chui, F.-T.; Kulkarni, V. R. WO Patent Appl.,
097,809, 2005.
9. Matteson, D. S. In Boronic Acids; Hall, D. G., Ed.; WILEY-VCH: Weinheim, 2005;
pp 305–342.
1
1
1
0. Matteson, D. S. Chem. Rev. 1989, 89, 1535–1551.
1. Matteson, D. S. Acc. Chem. Res. 1988, 21, 294–300.
2. Ryakhovsky, V. V.; Khachiyan, G. A.; Kosovova, N. F.; Isamiddinova, E. F.; Ivanov,
A. S. Beilstein J. Org. Chem. 2008, 4. doi:10.3762/bjoc.4.39
13. Baran, P. S.; Maimone, T. J.; Richter, J. M. Nature 2007, 446, 404–408. doi:10.
1038/nature05569
ꢀ
crystallized upon standing, mp 58–59.5 C. [Found: C, 67.30; H,
14. Barrett, G. C.; Elmore, D. T. Amino Acids and Peptides; Cambridge University
2
0
7.62; N,10.59. C29
4 4 D
H39BN O requires C, 67.18; H, 7.58; N,10.81.] [a]
Press: Cambridge, 1998.
ꢁ
40.0 (c 1.0, MeOH);
ESI-MS) 519.8 (MH ), 1038.1 (2MH );
n
max (KBr): 1518, 1685, 1714, 3265, 3386; m/z
15. El-Abadelah, M. M.; Sabri, S. S.; Jarrar, A. A.; Zarga, M. H. A. J. Chem. Soc., Perkin
Trans. 1 1979, 2881–2885. doi:10.1039/P19790002881
þ
þ
(
d
H
(360 MHz, acetone-d
6
)
16. Pecol, T.; Daniels, J. S.; Labutti, J.; Parsons, I.; Nix, D.; Baronas, E.; Hsieh, F.; Gan,
9
.20 (d, 1H, J 1.44 Hz, CHpyrazine), 8.82 (d, 1H, J 2.2 Hz, CHpyr-
L.-S.; Miwa, G. Drug Metab. Dispos. 2005, 33, 771–777.
azine), 8.63 (m, 1H, CHpyrazine), 8.40 (br d, 1H, NH), 7.61 (br s, 1H,
17. Shenvi, A. B. U.S. Patent 4,537,773, 1985.

 Ivanov, Andrey S.
Ivanov, Andrey S.
 Zhalnina, Anna A.
Zhalnina, Anna A.
 Shishkov, Sergey V.
Shishkov, Sergey V.