Vinylcyclopropane Epimerization
catalyst PhCHdRuCl2(PCy3)2 (3) (2.5, 5.0, or 7.5 mol %, 0.025 M
stock solution in toluene) or ethylidene catalyst2 MeCHdRuCl2-
(PCy3)2 (33) was added, and timing was initiated. Samples (100
µL) were withdrawn at 5 min intervals and analyzed by HPLC vs
external standards of starting compound (8) and its epimer (9). The
concentrations of 8 and 9 were determined against quantitative
HPLC calibration curves. Each experiment was repeated 1-5 times.
Each rate constant represents the average of 2-6 determinations.
NMR Kinetics. A. Grubbs Catalyst PhCHdRuCl2(PCy3)2 (3).
(1R,2S)-Methyl 1-benzoylamino-2-vinylcyclopropane carboxylate
(8) (36.9 mg, 0.15 mmol), internal standard 1,3,5-trimethoxybenzene
(26.8 mg, 99%, 0.158 mmol), and Grubbs catalyst PhCHdRuCl2-
(PCy3)2 (3) (12.3 mg, 0.015 mmol) were added to a dry vial under
Ar. CD2Cl2 (3.00 mL, degassed with Ar bubbling through at rt for
1 h) was added to dissolve the three components, and the resulting
purple homogeneous solution was transferred to an NMR tube with
a screw cap and septum under Ar. The sample was monitored by
Finally, tandem RCM-epimerization, as exemplified in the
(undesired, in this case) production of macrocycle 5, could be
exploited as a synthetically useful operation. It is hoped that
our mechanistic insight may help to plan such synthetic
operations. We are currently pursuing these and other strategies.
Experimental Section
General. All experiments were conducted under Ar or N2 atmos-
phere. Anhydrous THF, CH2Cl2, and toluene were degassed with
Ar before use. All of the reagents and catalysts used in this paper
were commercially available and used as received. (1R,2S)-1-Amino-
2-vinylcyclopropane carboxylic acid methyl ester tosylate salt (99%
ee) and its tert-butyloxycarbonyl derivatives were prepared as
previously described.11 The ethylidene species MeCHdRuCl2-
(PCy3)2 (33) was prepared according to the literature procedure.15
NMR Studies. Representative Example. (1R,2S)-Methyl 1-ben-
zoylamino-2-vinylcyclopropane carboxylate (8) (12.3 mg, 0.05
mmol) and Grubbs catalyst (3) (12.3 mg, 0.015 mmol) were added
to a dry vial under Ar. CD2Cl2 (1.00 mL, degassed with Ar bubbling
through at rt for 1 h) was added to dissolve the two compounds,
and a purple homogeneous solution resulted, which was then trans-
ferred to an NMR tube with a screw cap and septum under Ar.
1
1
NMR at 23.5 ( 0.5 °C (500.1 MHz H; 202.5 MHz 31P). H and
31P NMR were recorded at 15-20 min intervals. The concentrations
of 8 and 9 were determined based on integrations against the internal
standard. Each experiment was repeated 1-2 times.
B. Ethylidene Catalyst MeCHdRuCl2(PCy3)2 (33). (1R,2S)-
Methyl 1-benzoylamino-2-vinylcyclopropane carboxylate (8) (12.3
mg, 0.05 mmol), internal standard 1,3,5-trimethoxybenzene (18.3
mg, 99%, 0.108 mmol), and ethylidene catalyst2 MeCHdRuCl2-
(PCy3)2 (33) (38.0 mg, 0.05 mmol) were added to a dry vial under
Ar. Degassed CD2Cl2 (1.00 mL) was added, and the resulting purple
homogeneous solution was stirred at rt for 10 min and evaporated
in Vacuo to dryness (∼10 min). Another 1.00 mL of degassed CD2-
Cl2 was added, followed by the addition of a Cy3P solution in
toluene (1.0 M; 0.05, 0.1, or 0.2 mL; corresponding to 1, 2, or 4
equiv of Cy3P relative to the substrate). The resulting homogeneous
mixture was transferred to an NMR tube with a screw cap and
septum under Ar. The sample was monitored by NMR at 23.5 (
1
The sample was monitored by NMR at 303 K (500.1 MHz, H;
202.5 MHz, 31P) until epimerization reached equilibrium (2-3 h).
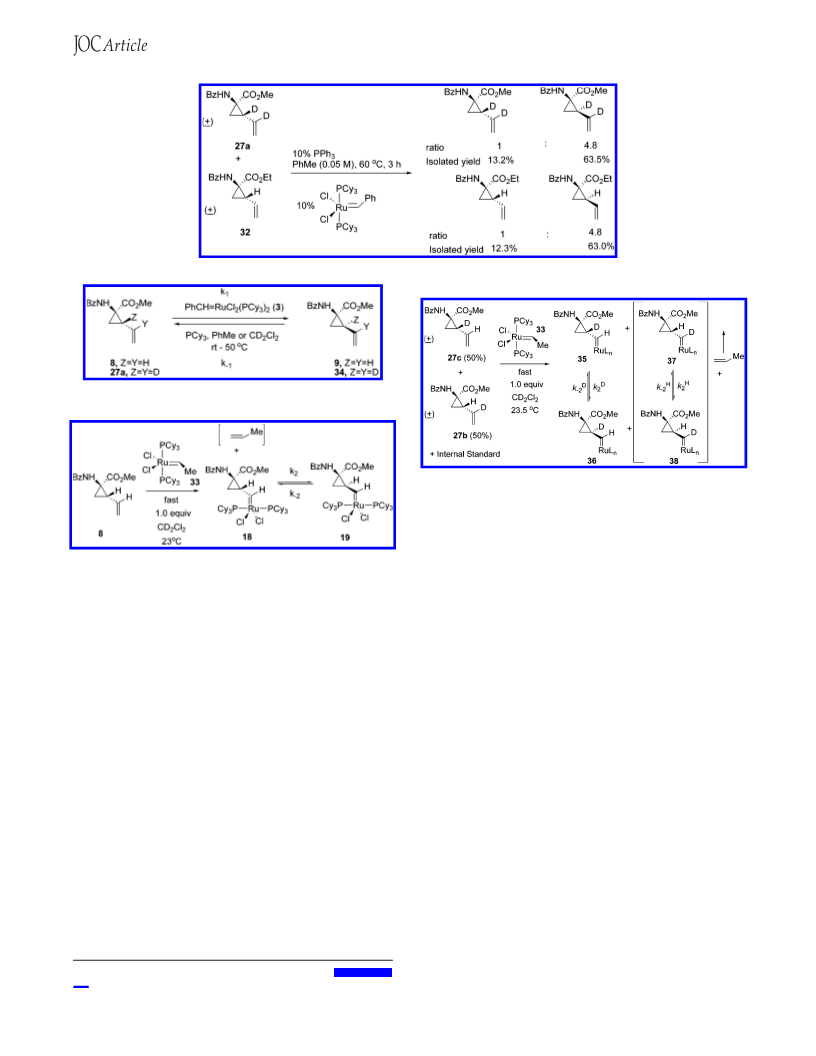
Crossover Study. A mixture of racemic methyl 1-benzoylamino-
2-deuterium-2-(1-deuteriumvinyl)cyclopropane carboxylate (27a)
(124 mg, 0.5 mmol), racemic ethyl 1-benzoylamino-2-vinylcyclo-
propane carboxylate (32) (130 mg, 0.5 mmol), Grubbs catalyst (41.1
mg, 0.05 mmol), and Ph3P (13.1 mg, 0.05 mmol) in toluene (20
mL) was stirred at 60 °C for 3 h and then concentrated. Flash
chromatography (silica gel, ethyl acetate/hexanes ) 2/3) gave recov-
ered racemic ethyl 1-benzoylamino-2-vinylcyclopropane carboxylate
(32) (16.0 mg, 12.3%), epi-racemic methyl 1-benzoylamino-2-
deuterium-2-(1-deuteriumvinyl)cyclopropane carboxylate (78.5 mg,
63.5%), and a mixture of racemic methyl 1-benzoylamino-2-deuter-
ium-2-(1-deuteriumvinyl)cyclopropane carboxylate (27a) and epi-
racemic ethyl 1-benzoylamino-2-vinylcyclopropane carboxylate.
The mixture was separated by preparative HPLC to give recovered
racemic methyl 1-benzoylamino-2-deuterium-2-(1-deuteriumvinyl)-
cyclopropane carboxylate (27a) (16.4 mg, 13.2%) and epi-racemic
ethyl 1-benzoylamino-2-vinylcyclopropane carboxylate (81.8 mg,
63.0%). Preparative HPLC conditions: mobile phases, water with
0.05% trifluoroacetic acid and acetonitrile with 0.05% trifluoroacetic
acid; column, Phenomenex Luna C18(2) 30 × 250, 10 µm; flow
rate, 25 mL/min; collection by UV at 220 nm. Identification data
for epi-racemic methyl 1-benzoylamino-2-deuterium-2-(1-deuteri-
umvinyl)cyclopropane carboxylate: 1H NMR (400 MHz, CDCl3)
δ 7.76-7.82 (m, 2H), 7.47-7.53 (m, 1H), 7.37-7.44 (m, 2H), 6.80
(bs, 1H), 5.28 (s, 1H), 5.20 (s, 1H), 3.69 (s, 3H), 2.00 (d, J ) 5.5
Hz, 1H), 1.40 (d, J ) 5.5 Hz, 1H); 13C NMR (100 MHz, CDCl3)
δ 172.02, 168.38, 134.07, 132.75 (t, J ) 24.2 Hz, 1C), 131.72,
128.42 (2C), 127.03 (2C), 118.45, 52.55, 38.74, 30.71 (t, J ) 24.2
Hz, 1C), 22.31. Identification data for epi-racemic ethyl 1-benzoy-
lamino-2-vinylcyclopropane carboxylate: 1H NMR (400 MHz,
CDCl3) δ 7.74-7.8 (m, 2H), 7.48-7.54 (m, 1H), 7.38-7.47 (m,
2H), 6.75 (bs, 1H), 5.57-5.66 (m, 1H), 5.31 (dd, J ) 17.1 and 0.9
Hz, 1H), 5.23 (dd, J ) 10.3 and 1.1 Hz, 1H), 4.16 (q, J ) 7.1,
2H), 2.50 (q, J ) 8.0 Hz, 1H), 2.04 (dd, J ) 10.5 and 5.4 Hz, 1H),
1.41 (dd, J ) 7.5 and 5.6 Hz, 1H), 1.23 (t, J ) 7.1 Hz, 3H).
Kinetic Studies. HPLC Kinetics. A mixture of (1R,2S)-Methyl
1-benzoylamino-2-vinylcyclopropane carboxylate (8) (123 mg, 0.5
mmol) and Cy3P (1.0 M in toluene, 0, 2.5, 5, 10, 15, or 20 mol %)
in toluene (degassed with Ar at 50 °C for 2 h, 0.01 M) was stirred
at 50 ( 0.2 °C under Ar for 30 min. A stock solution of Grubbs
1
1
0.5 °C (500.1 MHz H; 202.5 MHz 31P). H and 31P NMR were
recorded at 15-20 min intervals. The concentrations of 8 and 9
were determined based on integrations against the internal standard.
Each experiment was repeated 1-4 times.
Kinetic Isotope Effects. The HPLC and NMR kinetics experi-
ments were performed following the above example except that
(1R,2S)-methyl 1-benzoylamino-2-vinylcyclopropane carboxylate (8)
was replaced with racemic methyl 1-benzoylamino-2-deuterium-
2-(1-deuteriumvinyl)cyclopropanecarboxylate(27a).TheNMRkinet-
ics experiments were also performed on (1R,2S)-methyl 1-benzoy-
lamino-2-vinylcyclopropane carboxylate (8) and an equimolar mixture
of racemic methyl 1-benzoylamino-2-(1-deuteriumvinyl)cyclopro-
pane carboxylate (27b) and racemic methyl 1-benzoylamino-2-
deuterium-2-vinylcyclopropane carboxylate (27c), using 1 equiv of
the ethylidene catalyst2 MeCHdRuCl2(PCy3)2 (33) instead of 10
mol % of the Grubbs catalyst PhCHdRuCl2(PCy3)2 (3).
Acknowledgment. The authors thank Dr. Youla Tsantrizos
(Boehringer Ingelheim, Laval) for sharing her unpublished RCM
results, Prof. Maurizio Persico (Scuola Normale Superiore, Pisa)
for useful advice with the DFT calculations, and Prof. Donna
Blackmond (Imperial College, London) for helpful suggestions
with the kinetic experiments.
Supporting Information Available: Complete refs 13 and 20.
Detailed experimental procedures and characterization of all
compounds, plus energies and coordinates for all ground states and
transition states described in the paper. This material is available
JO061587O
J. Org. Chem, Vol. 71, No. 23, 2006 8875

 Zeng, Xingzhong
Zeng, Xingzhong
 Wei, Xudong
Wei, Xudong
 Farina, Vittorio
Farina, Vittorio
 Napolitano, Elio
Napolitano, Elio
 Xu, Yibo
Xu, Yibo
 Zhang, Li
Zhang, Li
 Haddad, Nizar
Haddad, Nizar
 Yee, Nathan K.
Yee, Nathan K.
 Grinberg, Nelu
Grinberg, Nelu
 Shen, Sherry
Shen, Sherry
 Senanayake, Chris H.
Senanayake, Chris H.