CATALYTIC METHOD OF SYNTHESIS OF PHOSPHORUS ACIDS ESTERS
1085
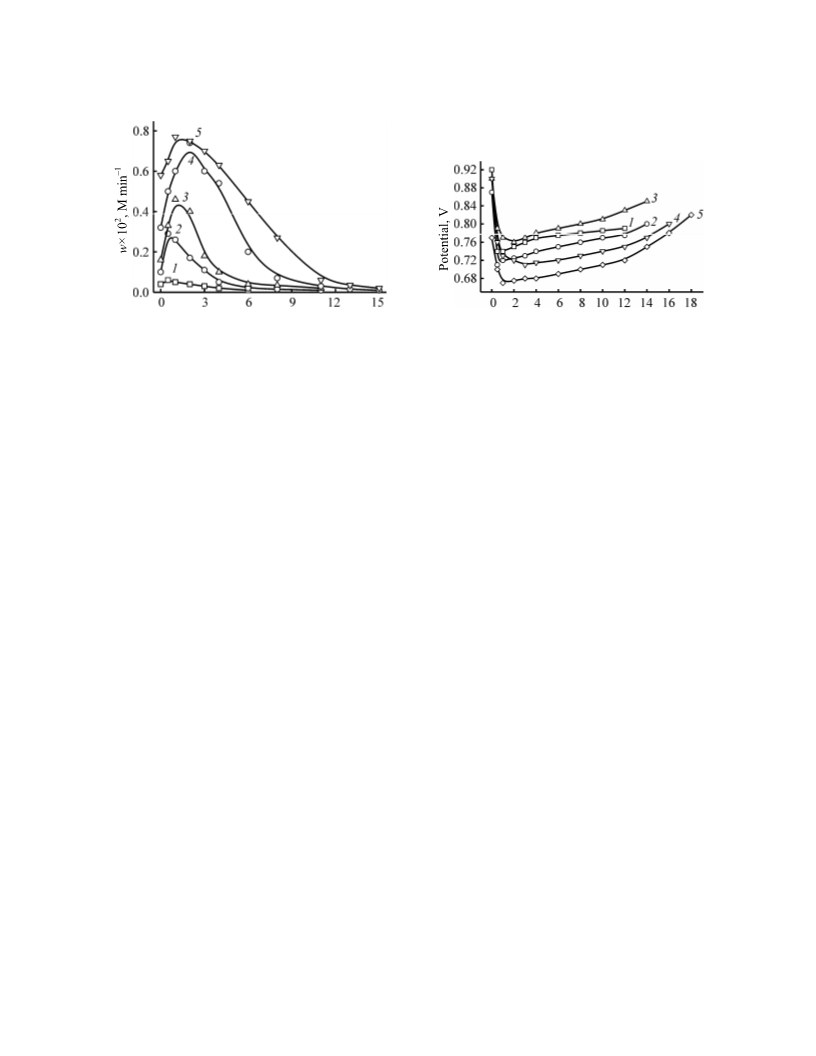
pressure 1 at, the molar ratio CuX2/Zn3P2 = 7–10,
HCl/Zn3P2 = 7–14. Copper bromide exhibits higher
catalytic activity and favors the formation of phos-
phoric acid ester. In the presence of copper chloride
dibutyl hydrogen phosphite and tributyl phosphate are
formed. The products were isolated from reaction
mixtures by vacuum distillation at the residual pressure
1–10 mm Hg and were identified. In 31P NMR spectra
the trialkyl phosphate is characterized by a signal with
the chemical shift 0±2 ppm, and dialkyl hydrogen
phosphite, by a signal at –8±2 ppm [9]. IR spectra of
obtained P(O)(OBu)3 contain typical absorption bands
of P=O (1260–1285 cm–1) and P–O–C (1020–1045 cm–1)
groups. In the IR spectra of P(O)H(OBu)2 the ab-
sorption band at 2430 cm–1 belongs to P–H bond
vibrations and a band of significantly smaller intensity
at 1260 cm–1, to the vibrations of phosphoryl group
P=O [10]. Absorption bands of the bending vibrations
of methylene groups at 1370–1470 cm–1 are also present.
Oxidative alcoholysis of zinc phosphide was
carried out in a closed system on an isothermal
installation consisting of the intensely shaken reactor
and the burette filled with oxygen. In the course of the
experiment Cu(II)/Cu(I) ratio was measured. For this
purpose the reactor was equipped with potentiometric
device consisting of platinum and calomel electrodes
with a joint playing the role of electrolytic key.
Experiments were carried out as follows. The reactor
was filled with butanol, then a catalyst was placed in it,
and the system was flushed with oxygen. After that a
weighed sample of zinc phosphide was introduced.
The total amount of solution in the reactor was 10–
15 ml. The amount of absorbed oxygen, the absorption
rate, and the current potential of reaction solution were
measured. The quantitative analysis of organo-
phosphorus products against the standard samples was
carried out on a Kristall 2000 M chromatograph
equipped with a flame ionization detector and a CP
SIL 19CB 25 m×0.25 mm capillary column after
removing copper salts from the sample by treating with
sodium bicarbonate. The conversion of phosphide was
calculated on the basis of the yield of organo-
phosphorus compounds. After the reduction of Cu(II)
to Cu(I) with thiosulfate and precipitating it with
iodide ion dibutyl hydrogen phosphite and tributyl
phosphate synthesized were isolated from reaction
mixture by vacuum distillation at 5–10 mm and
identified by IR and 31P NMR spectroscopy.
Hence, by the use of catalysts it is possible to
obtain organic phosphates and phosphites directly from
low-valent phosphorus compounds under mild condi-
tions. Harmless oxidation with oxygen can be used
instead of oxidation with toxic chlorine. Catalytic ap-
proach developed by us may be also used for
performing direct reactions of transition metal phos-
phides not only with alcohols, but also with the other
organic substrates.
EXPERIMENTAL
The work was carried out at the support of
ISTC#K-1284.2 program.
Environmentally safe oxygen was used as an
oxidant. It is often used in organic and inorganic
reactions with the participation of copper compounds
as catalysts [11].
1H and 31P NMR spectra were taken on a Bruker
Avance II 300 NMR spectrometer. IR spectra were
obtained on a Shimadzu IR prestige 21 Fourier IR
spectrometer.
REFERENCES
1. Tekhnologiya fosfora (Phosphorus Technology), Belov, V.N.,
Bol’shakova, A.P., and Dantsis, Ya.B., Eds., Leningrad:
Khimiya, 1979.
2. Chernogorenko, V.B., Novoe v poluchenii i primenenii
fosfidov i fosforsoderzhashchikh splavov (New Aspects
of Obtaining and Use of Phosphides and Phosphorus-
Containing Melts), Alma-Ata: Nauka, 1988, vol. 1, p. 5.
Purity of starting zinc phosphide was evaluated by
the emission spectral analysis on a DFS-13 device. It
was found that the content of impurities was no more
than 1%. Granulometric composition and surface of
zinc phosphide was evaluated by a Coulter register
(TAII model). Particles with the diameter 13.7 μcm
prevailed in zinc phosphide sample, and its specific
surface was 962 cm2 g–1. Preliminary drying of butanol
was carried out by boiling over CaO and subsequent
distillation. Oxygen-argon mixture was dried by
passing through calcium chloride.
3. Meyer, T., Honle, W., and Von Schnering, H.G.,
Z. Anorg. Allg. Chem., 1987, vol. 552, p. 69.
4. Trofimov, B.A., Brandsma, L., Arbuzova, S.N., Maly-
sheva, S.F., and Gusarova, N.K., Tetrahedron Lett.,
1994, vol. 35, no. 41, p. 7647.
5. Trofimov, B.A., Arbuzova, S.N., and Gusarova, T.N.,
Usp. Khim., 1999, vol. 68, no. 3, p. 240.
6. Ibraimova, Zh. U., Polimbetova, G.S., Aleshkova, M.M.,
and Borangazieva, A.K., Zh. Fiz. Khim., 2004, vol. 78,
no. 8, p. 1443.
RUSSIAN JOURNAL OF GENERAL CHEMISTRY Vol. 83 No. 6 2013

 Polimbetova
Polimbetova
 Ibraimova
Ibraimova
 Borangazieva
Borangazieva