New Journal of Chemistry
Please do not adjust margins
Page 10 of 12
Journal Name
97 39
ARTICLE
1
2
3
4
.
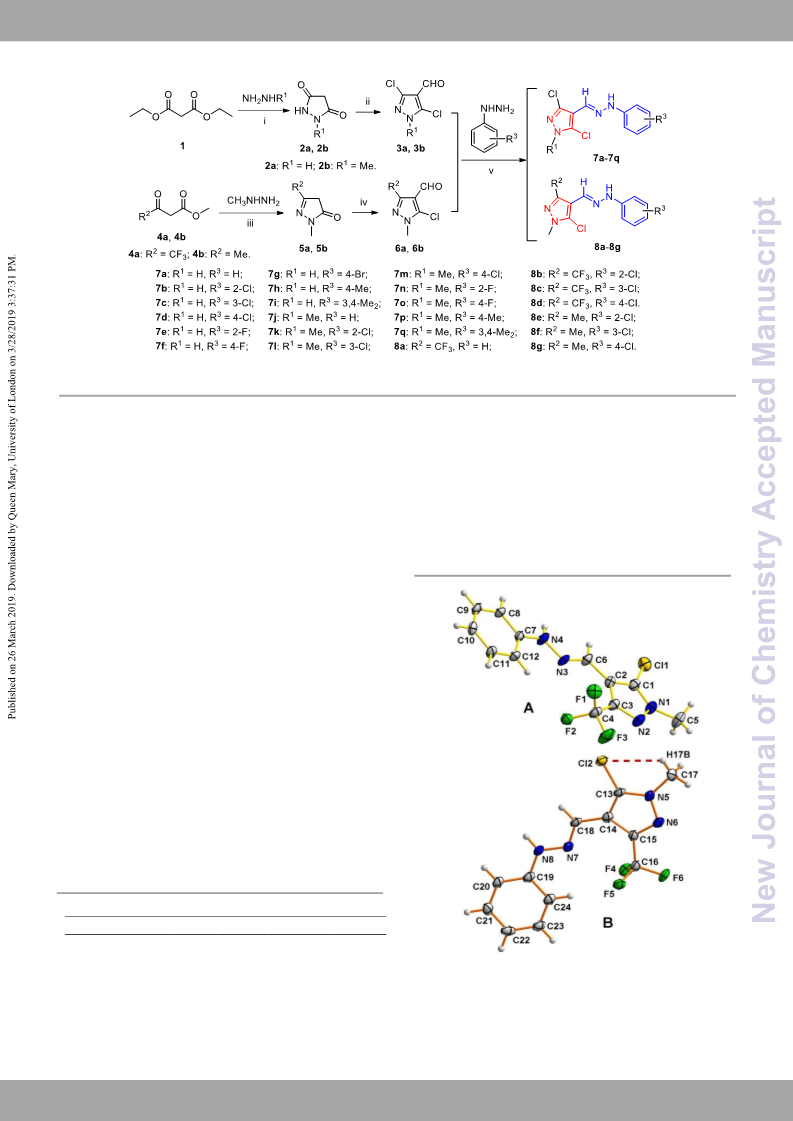
The X-ray data have been deposited at the Cambridge
There are no conflicts of interest to declare.
DOI: 10.1039/C9NJ00574A
Crystallographic Data Centre with the CCDC number 1846970
.
5
6
Acknowledgements
Antifungal activity assay
The authors gratefully acknowledge the financial support of
the National Natural Science Foundation of China (No.
31772209) and the Fundamental Research Funds for the
Central Universities of China (No. KYTZ201604).
7
8
9
The target compounds were tested for antifungal acitivity
against F. graminearum, B. cinerea and R. solani in vitro by
mycelium growth rate method.35,40 The tested fungus strains
were provided by the Laboratory of Plant Disease Control at
Nanjing Agricultural University. Every sample was dissolved in
DMSO and mixed with PSA (potato sucrose agar) medium. An
equal dose of DMSO in medium was used as the blank control.
Meanwhile, the commercial agricultural fungicides
carbendazim and penthiopyrad were tested as the positive
control at the same concentration. Each 15 mL medium was
poured into a 9 cm petri plate with 3 replicates. The fungi were
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
53
54
55
56
57
58
59
60
Notes and references
1. Z. Li, S. Chen, S. Zhu, J. Luo, Y. Zhang, Q. Weng, Molecules, 2015,
20, 13941–13957.
2. R. A. Wilso, N. J. Talbot, Microbiology, 2009, 155, 3810–3815.
3. G. Bai, G. Shaner, Annu. Rev. Phytopathol., 2004, 42, 135–161.
4. N. A. Foroud, F. Eudes, Int. J. Mol. Sci., 2009, 10, 147–173.
5. J. N. Sangshetti , D. B. Shinde, Bioorg. Med. Chem. Lett., 2010,
20, 742–745.
inoculated to the center of the medium and cultured at 25
℃
for 3–5 days in a dark environment. After a certain incubation
period, the diameters of the mycelium colonies were
measured, and the inhibitory percentages were calculated.
The median effective concentration (EC50) values were
calculated using linear-regression analysis.
6. F. Borges, F. Roleira, N. Milhazes, L. Santana, E. Uriarte, Curr.
Med. Chem., 2005, 12, 887–916.
7. K. D. Wehrstedt, P. A. Wandrey, D. Heitkamp, J. Hazard. Mater.,
2005, A126, 1–7.
8. P. Dai, K. Luo, X. Yu, W. -C. Yang, L. Wu, W. -H. Zhang, Adv. Synth.
Catal., 2018, 360, 468–473.
9. Y. Wang, Y. -P. Hou, C. -J. Chen, M. -G. Zhou, Australas. Plant
Path., 2014, 43, 307–312.
10. G. Bai, G. Shaner, Plant Disease, 1994, 78, 760–766.
11. M. F. El Shehry, M. M. Ghorab, S. Y. Abbas, E. A. Fayed, S. A.
Shedid, Y. A. Ammar, Eur. J. Med. Chem., 2018, 143, 1463–1473.
Med. Chem., 2014, 78, 86–96.
3D-QSAR study method
The
3D-QSAR model of target compounds was established via
built-in QSAR software of Sybyl X-2.1
(Tripos). The energy
minimization of target molecular conformations was
performed using the Tripos force field and the Gasteiger-
Hückel method with the 1000 times iteration and 0.005 kcal
1
1
mol– Å– ) convergence gradient. The fields properties of both
CoMFA and CoMSIA model were calculated using partial least
squares analysis (PLS).35,41
13. C. B. Vicentini, C. Romagnoli, E. Andreotti, D. Mares, J. Agric.
Food. Chem., 2007, 55, 10331–10338.
14. H. Dai, G. Li, J. Chen, Y. Shi, S. Ge, C. Fan, H. He, Bioorg. Med.
Chem. Lett., 2016, 26, 3818–3821.
15. H. Yu, Y. Cheng, M. Xu, Y. Song, Y. Luo, B. Li, J. Agric. Food. Chem.,
2016, 64, 9586–9591.
16. M. Bhat, G. K. Nagaraja, R. Kayarmar, S. K. Peethamber,
R. Mohammed Shafeeulla, RSC Advances, 2016, 6, 59375–
59388.
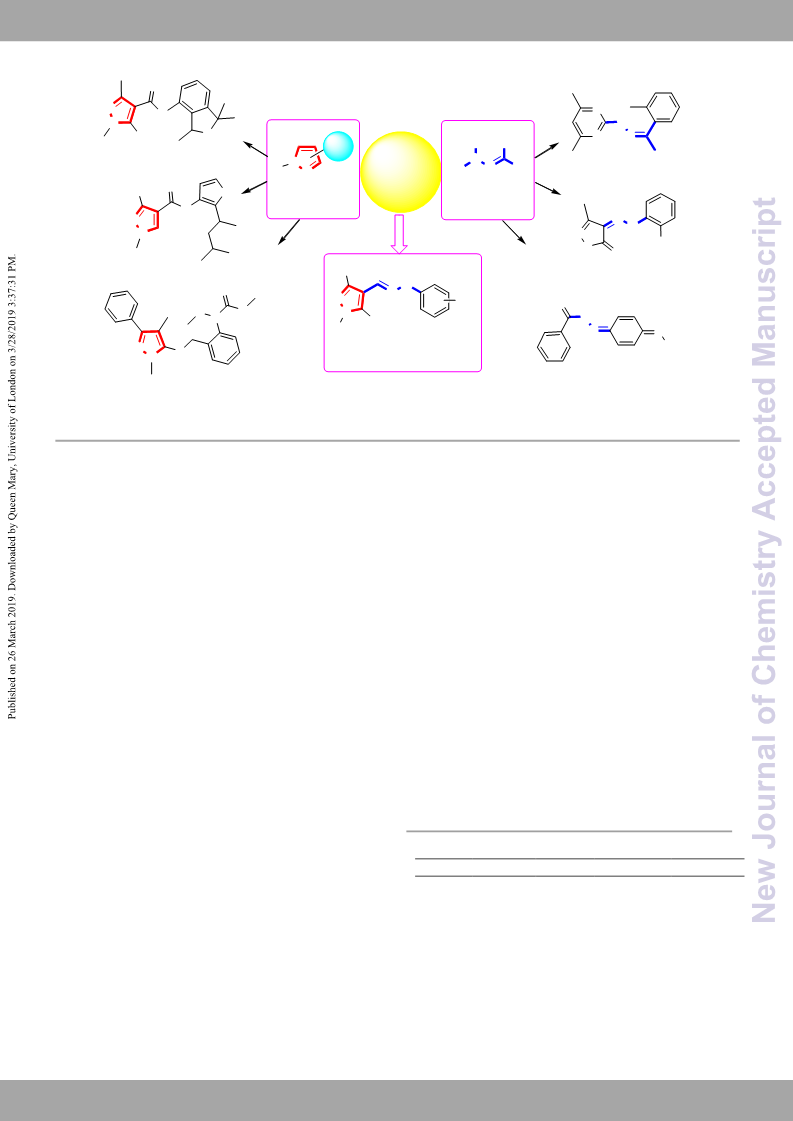
Conclusions
The substituted phenylhydrazone moieties were introduced to
4-position of 5-chloro-pyrazoles to design and synthesize a
series of novel pyrazole derivatives as potent antifungal agents.
The target compounds were well characterized by the
spectroscopy and single crystal X-ray diffraction. The bioassay
results indicated that most of target compounds exhibited
obvious fungicidal activity against the tested plant pathogenic
fungi. The compounds 7c and 8d showed most potent
inhibitory activity with the EC50 values of 0.68 μg/mL and 0.25
μg mL–1 against B. cinerea and R. solani, respectively, which are
superior than the corresponding control drugs carbendazim
and penthiopyrad. The CoMFA and CoMSIA models were
established for 3D-QSAR study, which showed good predictive
ability with satisfactory q2 and r2 values. The obtained analytic
results from the molecular models provided useful
information for further structural modification and
17. Y. Fu, M. -X. Wang, D. Zhang, Y. -W. Hou, RSC Advances, 2017,
7, 46858–46865.
18. H. Nagahori, H. Yoshino, Y. Tomigahara, N. Isobe, H. Kaneko, I.
Nakatsuka, J. Agric. Food Chem., 2000, 48, 5754–5759.
19. Y. Yoshikawa, H. Katsuta, J. Kishi, Y. Yanase, J. Pestic. Sci., 2011,
36, 347–356.
20. L. E. Gomez, L. Srigiriraju, A. Roy, WO 2017086970, 2017-05-26.
21. Z. Zhuang, N. Xu, Z. Zhuang, Y. Liu, Y. Han, J. Fan, Agrochemicals,
2016, 55, 316–319.
22. L. -M. Hu, X. -S. Li, Z. -Y. Chen, Z. -J. Liu, Chinese J. Org. Chem.,
2003, 23, 1131–1134.
23. L. -M. Yang, S. -B. Hua, Z. -J. Liu, Chinese J. Appl. Chem., 2005,
22, 829–834.
24. C. -X. Tan, D. -L. Shen, J. -Q. Weng, N. -B. Sun, X. -M. Ou, Chinese
J. Pestic. Sci., 2006, 8, 363–366.
25. J. Wu J, Q. Shi, Z. Chen, M. He, L. Jin, D. Hu, Molecules, 2012, 17,
5139–5150.
optimization of 5-chloro-pyrazole derivatives with
a
phenylhydrazone moiety to screen new fungal inhibitors with
high efficacy.
26. K. Matsuura, Y. Ishida, T. Kuragano, K. Konishi, J. Pestic. Sci.,
1994, 19, S197–S207.
27. I. F. Eckhard, K. Lehtonen, T. Staub, L. A. Summers, Aust. J.
Chem., 1973, 26, 2705–2710.
Conflicts of interest
This journal is © The Royal Society of Chemistry 20xx
J. Name., 2013, 00, 1-3 | 10
Please do not adjust margins

 Jiao, Jian
Jiao, Jian
 Wang, An
Wang, An
 Chen, Min
Chen, Min
 Wang, Meng-Qi
Wang, Meng-Qi
 Yang, Chun-Long
Yang, Chun-Long