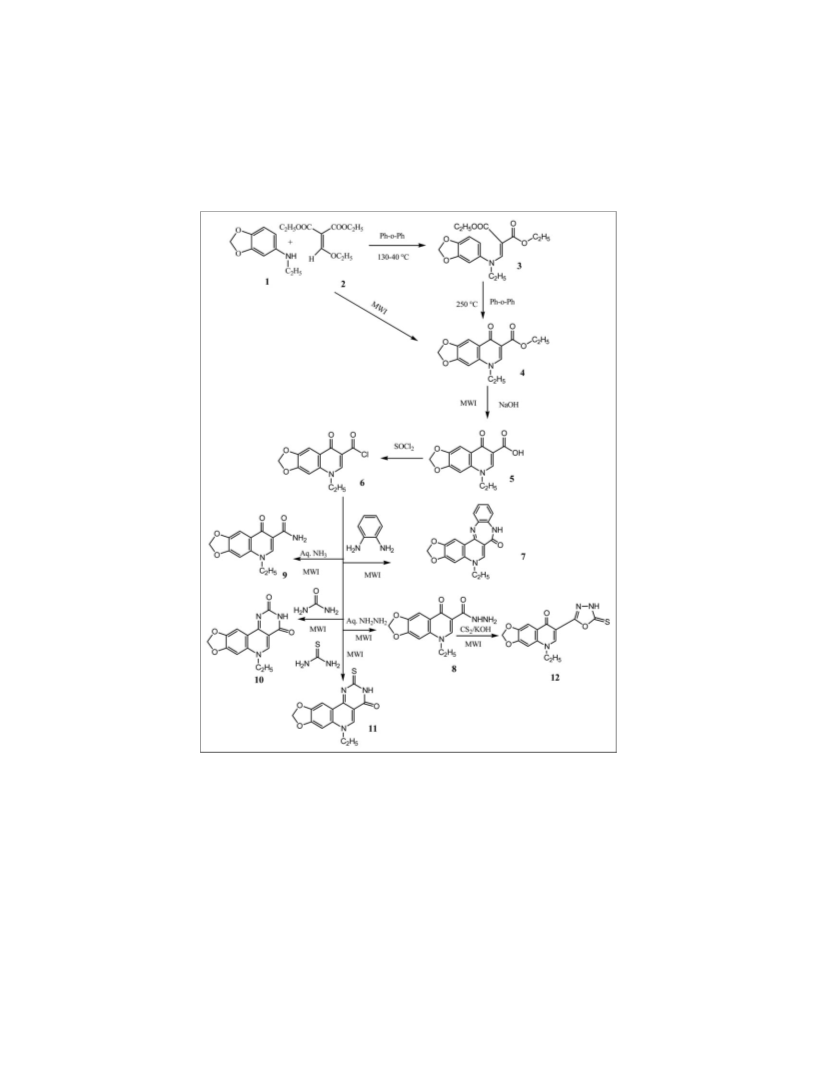
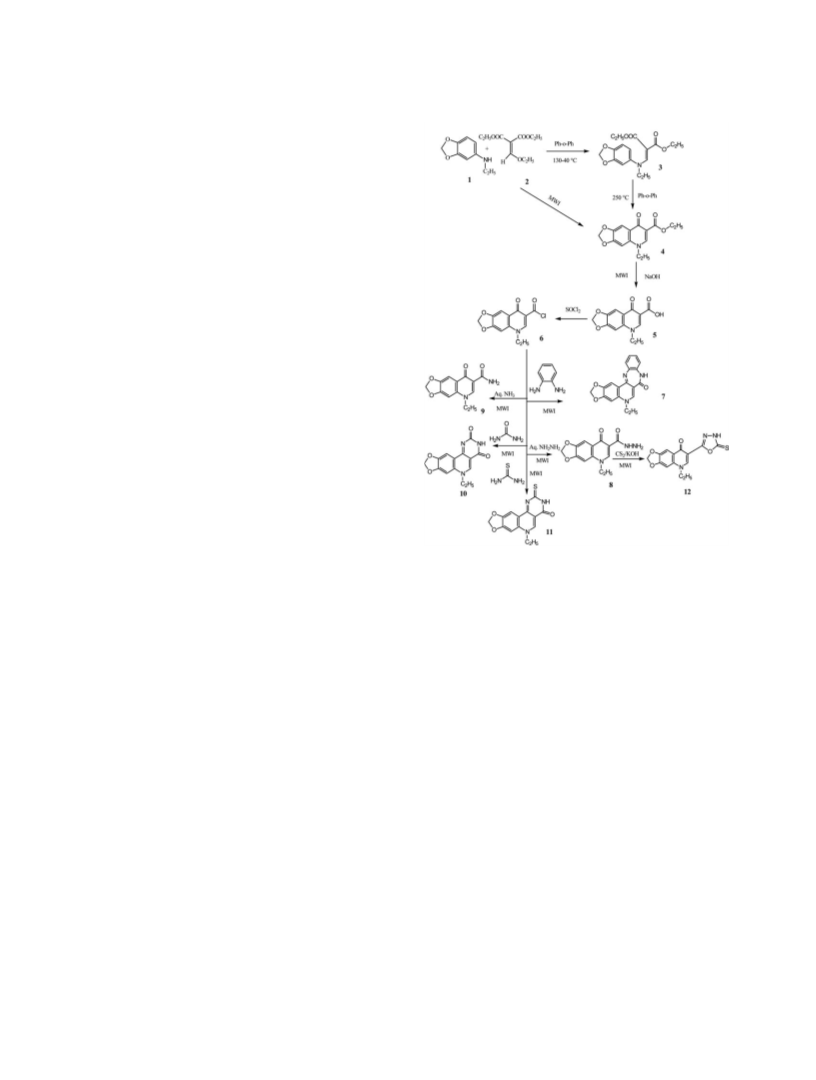
September 2010 Microwave-Assisted Synthesis of Quinolone Derivatives and Related Compounds
1107
reaction, as monitored by TLC the product was extracted into
ethanol (3 ꢁ 5 mL). Removal of solvent under reduced pres-
sure yielded the product, which was recrystallized from etha-
nol to give 0.38 g (92%) of 8 as yellow solid.
1037, 1017 cmꢂ1 1H NMR (DMSO-d6): d 1.42 (t, 3H), 4.66
;
(q, 2H), 6.35 (s, 2H), 7.74 (s, 1H), 8.26 (s, 1H), 9.11 (s, 1H),
15.62 (s,1H); MS (ESI, m/z):302[MþH]þ.Anal.Calcd. For
C14H11N3O3S: C, 55.80; H, 3.68; N, 13.95. Found: C, 55.70;
H, 3.88; N, 14.05.
mp 300–302ꢀC; IR (KBr): 3400, 3245, 2986, 1647, 1618,
1592, 1506, 1474, 1375, 1269, 1256, 1238, 1197, 1038 cmꢂ1
;
5-Ethyl -7-(5-thioxo-4, 5-dihydro-[1, 3, 4] oxadiazol-2-yl)-
5-H-[1, 3] di-oxolo [4, 5-g] quinolin-8-one (12).
MS (ESI, m/z):276 [MþH]þ. Anal. Calcd. For C13H13N3O4: C,
56.72; H, 4.76; N, 15.27. Found: C, 56.87; H, 4.56; N, 15.54.
5-Ethyl-8-oxo-5,8-dihydro-[1,3]dioxolo[4,5-g] quinoline-7-
carboxamide (9). Method A. This compound was obtained
in 77% yield as a white solid (ref. [19]).
Method
A. A mixture of 8 (0.30 g, 0.0011 mol), CS2 (0.68 g, 0.009
mol), and KOH (0.22 g, 0.004 mol) in ethanol 30 mL was
heated under reflux for 10 h, cooled to room temperature, and
diluted with water (4 mL) was heated under reflux until the
evolution of H2S had ceased. The reaction mixture was cooled
to room temperature, and poured into water and acidification
with HCl. The resulting precipitated was filtered and washed
with water and recrystallized from methanol to give 0.25 g
(71%) of 12.
Method B. To a solution of (8) (0.30 g, 0.0011 mol), CS2
(0.68 g, 0.009 mol) and KOH (0.22 g, 0.004 mol) in ethanol
20 mL dissolved in water (4 mL) were added. The reaction
mixture was subjected to microwave irradiation in a Pyrex
glass round bottle flask attached with water condenser from
outside with special arrangement for 4 min (Table 1). On com-
pletion of reaction, as monitored by TLC the reaction mixture
was cooled to room temperature and poured into water and
acidification with HCl. The resulting precipitated was filtered
and washed with water and recrystallized from methanol to
give 0.3 g (86%) of 12.
mp 260–263ꢀC; IR (KBr): 3442, 3171, 3050, 2901,
1650,1621, 1595, 1519, 1483, 1392, 1269, 1255, 1230,
1197,1039 cmꢂ1 1H NMR (DMSO-d6): d 1.35 (t, 3H), 4.40
;
(q, 2H), 6.19 (s, 2H), 7.48 (s, 1H), 7.52 (s, 1H) 8.58 (s, 1H),
11.26 (s, 1H); MS (ESI, m/z):318 [MþH] þ. Anal.Calcd. for
C14H11N3O4S: C, 52.99; H, 3.49; N, 13.24. Found: C, 52.88;
H, 3.36; N, 13.54.
Caution: Although we did not have any accident in this
work, it is highly recommended that the reaction should be
performed in an efficient hood.
In vitro antibacterial activity. Minimal inhibitory concen-
trations (MICs) were determined by means of an agar dilution
method, using Mueller Hinton agar plates containing a series
of twofold dilutions of drug. Overnight cultures in Mueller
Hinton broth were used for precultures of tested strains. MICs
were determined after overnight incubation at 37ꢀC, with an
inoculum equivalent to 1:100 dilution of an 18 h culture in
Mueller Hinton broth (about108 cells/mL). Each inoculum was
seeded onto agar plates by using an inoculum-replicating appa-
ratus and transferred by a 0.005 mL (about 104 cells) cali-
brated loop. The MIC was the lowest drug concentration that
inhibited the development of visible growth on agar plate.
The in vivo antibacterial activities of drugs were determined
in the systemic infections of mice with bacteria. Ten female
Swiss albino mice weighing 18 to 22 g were used for each
dose level. Microorganisms grown on Mueller Hinton agar
plates were suspended in physiological saline solution. Mice
were intraperitoneally challenged with bacteria. Mice infected
with Escherichia coli ML 4707 were treated at 1 h after infec-
tion. Mice infected with Klebsiella pneumoniae ML 4730 and
Proteus morganii ML 4731 were treated, respectively, at 1, 6,
and 24 h and then immediately and at 3 h after infection.
Drugs to be tested were administered PO. The total number of
surviving mice was recorded 1 week after infection, and the
Method B. To a solution of 6 (0.28 g, 0.001 mol), ammonia
solution (5 mL) in ethanol, basic alumina (2 g) was added.
The reaction mixture was stirred well, air dried, and subjected
to MWI intermittently at 30 s intervals for specified time
(Table 1). On completion of reaction, as monitored by TLC
the product was extracted into ethanol (3 ꢁ 5 mL). Removal
of solvent under reduced pressure yielded the product which
was recrystallized from ethanol to give 0.24 g (92%) of 9 as a
white solid. mp 334–336ꢀC; (lit. ref. [19] 336ꢀC).
6-Ethyl-6H-8, 10-dioxa-1, 3, 6 triaza-cyclopenta [b] phen-
anthrene-2, 4-dione (10). Method A. A mixture of 6 (0.28
g, 0.001 mol), urea (0.1 g, 0.002 mol), 5% aq. KOH (2 mL)
and methanol (10 mL) was refluxed for 2 h and cooled. The
precipitate obtained after dilution with water was filtered,
washed with water, and recrystallized from methanol to give
0.20 g (70%) of 10 as a white solid.
Method B. To a solution of compound 6 (0.28 g, 0.001
mol), urea (0.1 g, 0.002 mol) in methanol (10 mL), 5% aq
KOH (2 mL), and basic alumina 2 g was added. The reaction
mixture was stirred well, air dried, and subjected to MWI
intermittently at 30 s intervals for specified time (Table 1). On
completion of reaction, as monitored by TLC the product was
extracted into ethanol (3 ꢁ 5 mL). Removal of solvent under
reduced pressure yielded the product which was recrystallized
from methanol to give 0.25 g (88%) of 10 as a white solid.
mp 285–287ꢀC; IR (KBr): 3431, 2926,2853,1721, 1685,
1658, 1604, 1574, 1557, 1508, 1472, 1416, 1335, 1292, 1251,
1216, 1181, 1114, 1084,1024 cmꢂ1 1H NMR (DMSO-d6): d
;
1.42 (t, 3H), 4.66 (q, 2H), 6.35 (s, 2H), 7.74 (s, 1H), 8.26 (s,
þ
1H), 9.11 (s, 1H), 15.62 (s, 1H); MS (ESI, m/z):286 [MþH]
.
Anal.Calcd. For C14H11N3O4: C, 58.95; H, 3.89; N, 14.73.
Found: C, 58.85; H, 3.95; N, 14.85.
6-Ethyl – 2- thioxo-2, 6 dihydro-3H-8, 10-dioxa-1, 3, 6 tri-
aza-cyclopenta [b] phenanthrene - 4-one (11). Method A. A
mixture of 6 (0.28 g, 0.001 mol), thiourea (0.15 g, 0.002 mol),
5% aq. KOH (2 mL), and methanol (10 mL) was refluxed for
2 h and cooled. The precipitate obtained after dilution with
water was filtered, washed with water, and recrystallized from
methanol to give 0.21 g (70%) of 11 as a white solid.
Method B. To a solution of 6 (0.28 g, 0.001 mol), thiourea
(0.15 g, 0.002 mol) in methanol (10 mL), 5% aq KOH 2 mL,
and basic alumina 2 g was added. The reaction mixture was
stirred well; air dried, and subjected to MWI intermittently at
30 s intervals or specified time (Table 1).On completion of
reaction, as monitored by TLC, the product was extracted into
ethanol (3 ꢁ 5 mL). Removal of solvent under reduced pres-
sure yielded the product, which was recrystallized from metha-
nol to give 0.26 g (86%) of 11 as a white solid.
mp 265–268ꢀC; IR (KBr): 3401, 3061, 2363, 1708, 1629,
1553, 1499, 1471, 1433, 1396, 1370, 1272, 1250, 1221, 1166,
Journal of Heterocyclic Chemistry
DOI 10.1002/jhet

 Pednekar, Suhas
Pednekar, Suhas
 Pandey, Anil Kumar
Pandey, Anil Kumar