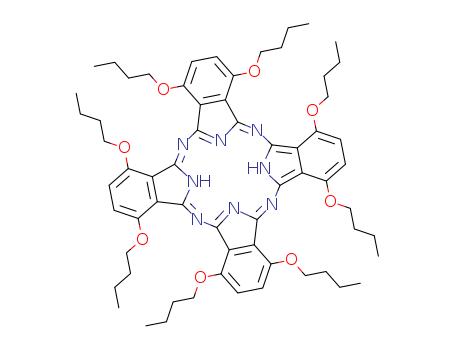
Chemical Property of 1,4,8,11,15,18,22,25-Octa-n-butoxyphthalocyanine
Chemical Property:
- Melting Point:221 °C (dec.)(lit.)
- Refractive Index:1.62
- PSA:179.58000
- Density:1.178g/cm3
- LogP:11.32400
- XLogP3:16.1
- Hydrogen Bond Donor Count:2
- Hydrogen Bond Acceptor Count:14
- Rotatable Bond Count:32
- Exact Mass:1090.62556160
- Heavy Atom Count:80
- Complexity:1610
- Purity/Quality:
-
98%,99%, *data from raw suppliers
1,4,8,11,15,18,22,25-Octabutoxy-29H,31H-phthalocyanine
Dye content 95 % *data from reagent suppliers
Safty Information:
- Pictogram(s):
- Hazard Codes:
- Safety Statements:
22-24/25
- MSDS Files:
-
SDS file from LookChem
Total 1 MSDS from other Authors
Useful:
- Canonical SMILES:CCCCOC1=C2C(=C(C=C1)OCCCC)C3=NC4=NC(=NC5=NC(=NC6=C7C(=CC=C(C7=C(N6)N=C2N3)OCCCC)OCCCC)C8=C(C=CC(=C85)OCCCC)OCCCC)C9=C(C=CC(=C94)OCCCC)OCCCC
-
General Description
1,4,8,11,15,18,22,25-Octabutoxyphthalocyanine is a phthalocyanine derivative with eight butoxy substituents positioned at the non-peripheral (α) positions of the macrocycle. This substitution pattern influences the electronic and structural properties of the phthalocyanine core, as evidenced by its use in studies involving axial ligand exchange for tuning near-IR absorption in manganese(III) phthalocyanines. The butoxy groups expand the central cavity of the phthalocyanine skeleton while reducing its ring current, which can affect spectroscopic behavior. Additionally, 1,4,8,11,15,18,22,25-OCTABUTOXY- PHTHALOCYANINE has been employed in the synthesis of double-decker terbium(III) complexes, where its protonation and deprotonation behavior were investigated, highlighting its role in modulating the coordination environment of the metal center.