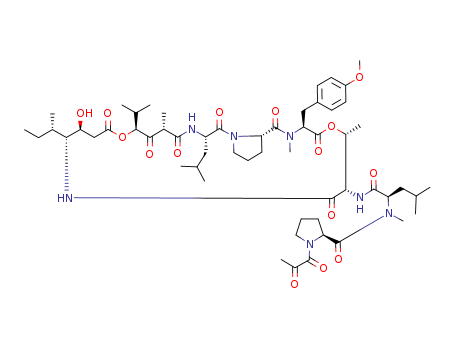
Multi-step reaction with 10 steps
1: 99 percent / DMAP, DCC / CH2Cl2 / 7 h / 0 °C
2: TFA / CH2Cl2 / 0.33 h
3: 76 percent / 1-hydroxybenzotriazole, N-<(1H-benzotriazol-1-yl)(dimethylamino)methylene>-N-methylmethanaminium hexafluorophosphate N-oxide, N,N-diisopropylethylamine / acetonitrile / 3 h / Ambient temperature
4: 78 percent / H2 / Pd/C / tetrahydrofuran / 1 h / 760 Torr / Ambient temperature
5: 60 percent / DMAP*CF3COOH, N,N'-diisopropylcarbodiimide / CHCl3 / 1.) reflux, 8 h, 2.) room temperature, 16 h
6: 81 percent / aq. NH4OAc, Zn / tetrahydrofuran / 24 h
7: 95 percent / H2 / Pd/C / tetrahydrofuran / 4 h / 760 Torr / Ambient temperature
8: 28 percent / N-<(dimethylamino)-1H-1,2,3-triazolo<4,5-b>pyridin-1-ylmethylene>-N-methylmethanaminium hexafluorophosphate N-oxide, 1-hydroxy-7-azabenzotriazole, N,N-diisopropylethylamine / tetrahydrofuran / 3 h / 0 °C
9: 98 percent / 5 M HCl / dioxane / 1 h
10: 62 percent / bromotripyrrolidinophosphonium hexafluorophosphate, diisopropylethylamine / CH2Cl2 / 1.) -5 deg C, 30 min, 2.) room temperature, 4 h
With
hydrogenchloride; dmap; 1-hydroxy-7-aza-benzotriazole; N-<(1H-benzotriazol-1-yl)(dimethylamino)methylene>-N-methylmethanaminium hexafluorophosphate N-oxide; N-<(dimethylamino)-1H-1,2,3-triazolo<4,5-b>pyridin-1-ylmethylene>-N-methylmethanaminium hexafluorophosphate N-oxide; ammonium acetate; hydrogen; benzotriazol-1-ol; 4-N,N-dimethylaminopyridine trifluoroacetate; N-ethyl-N,N-diisopropylamine; dicyclohexyl-carbodiimide; bromo-tris(1-pyrrolidinyl)phosphonium hexafluorophosphate; trifluoroacetic acid; diisopropyl-carbodiimide; zinc;
palladium on activated charcoal;
In
tetrahydrofuran; 1,4-dioxane; dichloromethane; chloroform; acetonitrile;
DOI:10.1021/jo961932h