Refernces
10.1002/anie.201308984
The research focuses on the development and systematic comparison of peptidic proteasome inhibitors, with a particular emphasis on the α-ketoamide electrophile as a promising reversible lead motif for therapeutic applications in oncological and immunological contexts. The study involves a series of in vitro, in vivo, and structural experiments to evaluate the implications of altered functionality and chemical reactivity of different electrophilic warheads on the inhibitory potential of the compounds. The researchers synthesized a range of inhibitors with varying electrophilic headgroups, including α-ketoaldehydes, α',β'-epoxyketones, boronic acids, and vinylsulfone, all based on the Z-Leu-Leu-Leu backbone. They performed IC50 measurements to assess the chymotrypsin-like activity of the proteasome, conducted crystallographic binding analysis to understand the binding profiles at the atomic level, and evaluated the cytotoxic effects in HeLa cell cultures. The experiments involved the use of various biochemical and structural methods, such as Grignard reactions, oxidation with 2-iodoxybenzoic acid (IBX), and synchrotron radiation for data recording. The analyses included IC50 measurements, Alamar Blue viability assays, and pulse chase experiments to study the kinetic behavior of the compounds. The results indicated that α-ketoamides, despite lacking a second strong electrophile, showed high binding affinities and selectivity for malignant tumor cells, suggesting their potential for expanded utility in chemo- and immunosuppressive therapies.
10.1021/jo00252a017
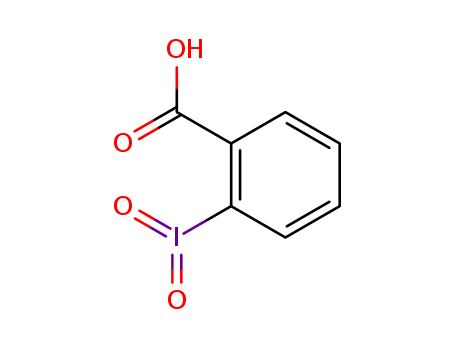
The research focuses on the synthesis and catalytic activity of 2-iodoso- and 2-iodoxybenzoic acids, which contain various substituents such as alkyl, alkyloxy, nitro, carboxyl, and water-solubilizing groups. The purpose of this study was to determine the influence of these substituents on the rates of hydrolysis of active phosphorus esters, including p-nitrophenyl diphenyl phosphate (PNPDPP), p-nitrophenyl isopropylphenylphosphinate (NPIPP), and p-nitrophenyl hexanoate (PNPH), in the presence of cetyltrimethylammonium chloride (CTAC). The research concluded that all synthesized compounds act as true catalysts, with their catalytic activity increasing with higher catalyst concentrations. Notably, 2-iodoxybenzoic acids showed 60-110% of the activity of their 2-iodosobenzoic acid analogues in 0.001 M CTAC. The study found that lipophilic substituents significantly enhanced the catalytic rates, while electron-releasing, -withdrawing, and water-soluble groups had moderate effects. The research also identified that 5-(2-hydroxyethoxy)-2-iodoxybenzoic acid and 5-(alkyloxy)-2-iodosobenzoic and -2-iodoxybenzoic acid derivatives exhibited extraordinary rate enhancements, making these compounds potentially useful as decontaminants for active phosphorus derivatives.
10.1039/c5ob02659h
The research aims to develop an efficient and environmentally friendly method for synthesizing iminoquinones, which are important in natural products and have diverse biological activities. The study introduces a chemoselective domino oxidative homocoupling of 2-aminoaryl ketones using 2-iodoxybenzoic acid (IBX) as the oxidant. The domino reaction involves three consecutive steps: ortho-hydroxylation of 2-aminoaryl ketones, oxidation of phenol derivatives to benzoquinones, and imine formation to yield iminoquinones. The researchers optimized the reaction conditions, finding that using 2 equivalents of IBX in DMSO at room temperature for 10 minutes provided the best results. The method allows for the recycling of IBX by converting the by-product iodosobenzoic acid (IBA) back to IBX. The study concludes that this domino strategy is highly chemoselective, efficient, and functional group tolerant, making it a viable approach for synthesizing iminoquinones from various substrates, including those containing sensitive functional groups.


 Xn
Xn




