10.1016/S0957-4166(98)00087-1
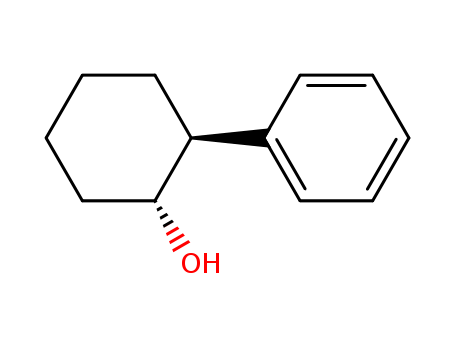
The research focuses on the asymmetric Diels–Alder addition of cyclopentadiene to chiral 1,4-naphthoquinones, with the aim of achieving high levels of diastereomeric excess. The purpose of this study was to develop a method for the stereoselective formation of cyclopentannulated products, which can be further transformed into pyranonaphthoquinones, a class of compounds related to the pyranonaphthoquinone antibiotics. The researchers used various chiral auxiliaries, including (R)-pantolactone, (S)-N-methyl-2-hydroxysuccinimide, and trans-2-phenylcyclohexanol, which when combined with Lewis acid conditions, led to significant asymmetric induction. The conclusions drawn from the study were that the use of chiral auxiliaries at C-2 of 1,4-naphthoquinones enabled up to 96% stereoinduction in Diels–Alder cycloadditions with cyclopentadiene. The chiral auxiliaries could be removed from the fragmented products in acceptable yields, allowing for the formation of cyclopentannulated pyranonaphthoquinone ring systems, similar to those found in nature.