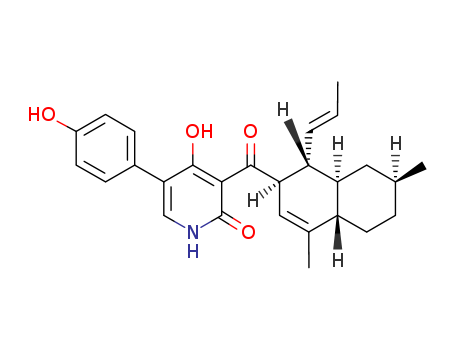
- Chemical Name:ilicicolin H
- CAS No.:12689-26-8
- Molecular Formula:C27H31 N O4
- Molecular Weight:433.547
- Hs Code.:
- Mol file:12689-26-8.mol
Synonyms:2(1H)-Pyridinone,4-hydroxy-5-(4-hydroxyphenyl)-3-[[(1R,2S,4aS,7S,8aR)-1,2,4a,5,6,7,8,8a-octahydro-4,7-dimethyl-1-(1E)-1-propenyl-2-naphthalenyl]carbonyl]-(9CI); 2(1H)-Pyridinone, 4-hydroxy-5-(4-hydroxyphenyl)-3-[[1,2,4a,5,6,7,8,8a-octahydro-4,7-dimethyl-1-(1-propenyl)-2-naphthalenyl]carbonyl]-,[1R-[1a(E),2b,4ab,7b,8aa]]-; Ilicicolin H;Illicicolin H