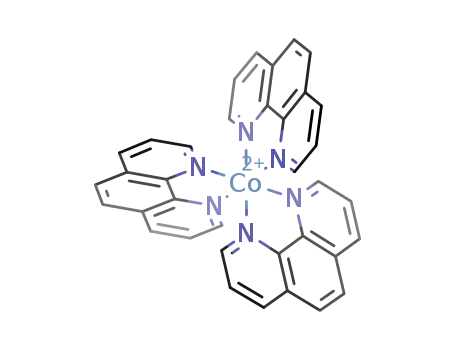
- Chemical Name:triphenanthrolinecobalt(III)
- CAS No.:16788-34-4
- Molecular Formula:C36H24 Co N6
- Molecular Weight:599.62
- Hs Code.:
- Mol file:16788-34-4.mol
Synonyms:Cobalt(2+),tris(1,10-phenanthroline)-, ion (8CI); Cobalt(2+),tris(1,10-phenanthroline-N1,N10)-, (OC-6-11)-; Cobalt(2+),tris(1,10-phenanthroline)-; Tris(1,10-phenanthroline)cobalt(2+)