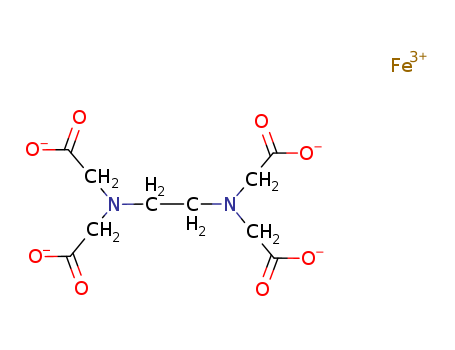
- Chemical Name:Iron(III)-edta
- CAS No.:15275-07-7
- Molecular Formula:C10H12FeN2O8
- Molecular Weight:344.061
- Hs Code.:
- DSSTox Substance ID:DTXSID201033262
- Wikipedia:Ferric_EDTA
- Wikidata:Q27113963
- Mol file:15275-07-7.mol
Synonyms:ammonium ferric edetate;EDTA Fe(III);EDTA ferric ammonium;Fe(III)-EDTA;Fe(III)-edta complex (1:1);Fe(III)-EDTA, ammonium salt;Fe(III)-EDTA, potassium salt;Fe(III)-EDTA, sodium salt;Fe(III)-EDTA, sodium salt, trihydrate;ferrate(1-), ((ethylenedinitrilo)tetraacetato)-, hydrogen;ferric EDTA;ferric sodium edetate;ferric-edta;hydrogen ((ethylenedinitrilo)tetraacetato)ferrate(III);iron(III) EDTA;Irostrene;monoferric edetate;NaFeEDTA;sodium feredetate;sodium iron EDTA;Sytron