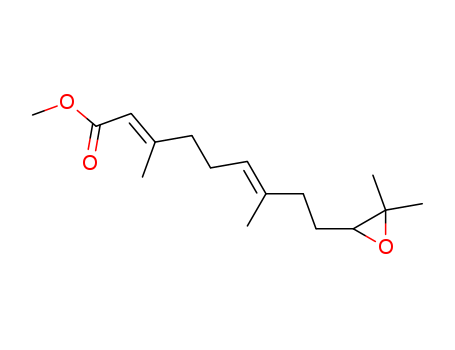
- Chemical Name:Juvenate
- CAS No.:24198-95-6
- Deprecated CAS:76898-57-2,34769-49-8,5299-11-6,20143-98-0
- Molecular Formula:C16H26 O3
- Molecular Weight:266.381
- Hs Code.:2918990090
- European Community (EC) Number:246-072-9
- NSC Number:688246
- DSSTox Substance ID:DTXSID601033769
- Nikkaji Number:J142.412B,J3.044.914B
- Metabolomics Workbench ID:125858
- ChEMBL ID:CHEMBL1967398
- Mol file:24198-95-6.mol
Synonyms:10,11-epoxyfarnesenic acid methyl ester;juvenile hormone III;juvenile hormone III, (E,E)-(+-)-isomer;juvenile hormone III, (E,E)-isomer;juvenile hormone III, (R-(E,E))-isomer;juvenile hormone III, (S-(E,E))-isomer;juvenile hormone III, (Z,E)-isomer;methyl 10,11-epoxy-3,7,11-trimethyl-2,6-dodecanoate