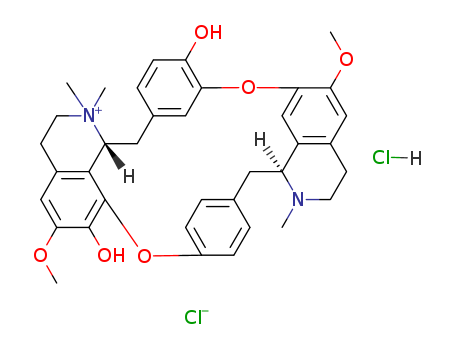
Chemical Property of Tubocurarine Chloride
Chemical Property:
- Appearance/Colour:CLEAR COLOURLESS LIQUID
- Melting Point:274~275℃
- Refractive Index:1.7350 (estimate)
- Boiling Point:°Cat760mmHg
- PKA:pK: 7.4(at 25℃)
- Flash Point:°C
- PSA:80.62000
- Density:g/cm3
- LogP:4.40370
- Storage Temp.:2-8°C
- Hydrogen Bond Donor Count:3
- Hydrogen Bond Acceptor Count:8
- Rotatable Bond Count:2
- Exact Mass:680.2419925
- Heavy Atom Count:47
- Complexity:990
- Purity/Quality:
-
(+)-Tubocurarine Chloride - CAS 57-94-3 - Calbiochem *data from reagent suppliers
Safty Information:
- Pictogram(s):
T
- Hazard Codes:T
- Statements:
25
- Safety Statements:
45
- MSDS Files:
-
SDS file from LookChem
Total 1 MSDS from other Authors
Useful:
- Canonical SMILES:CN1CCC2=CC(=C3C=C2C1CC4=CC=C(C=C4)OC5=C6C(CC7=CC(=C(C=C7)O)O3)[N+](CCC6=CC(=C5O)OC)(C)C)OC.Cl.[Cl-]
- Isomeric SMILES:CN1CCC2=CC(=C3C=C2[C@@H]1CC4=CC=C(C=C4)OC5=C6[C@@H](CC7=CC(=C(C=C7)O)O3)[N+](CCC6=CC(=C5O)OC)(C)C)OC.Cl.[Cl-]
-
Description
The name curare is derived from the native Guyana Mukusi
Indian word wurari. In 1596, Sir Walter Raleigh referred to
curare in The Discovery of the Large, Rich, and Beautiful Empire of
Guiana. In 1780, Abbe Felix Fontana identified the action of
curare on voluntary muscles. In 1800, Alexander von Humboldt
described the extraction of curare. In 1811, Sir Benjamin
Collins Brodie determined that complete recovery from curare
poisoning is possible provided artificial ventilation is maintained.
In 1825, Charles Waterton brought curarep to Europe,
and in 1835 Sir Robert Hermann Schomburgk classified and
named the vine Strychnos toxifera. In 1850, George Harley
demonstrated that curare could be used to treat tetanus and
strychnine poisoning. By 1868, Claude Bernard and Alfred
Vulpian had identified the site of action of curare as the motor
end plate. From 1887, curare was marketed for medical use by
Burroughs Welcome. In 1900, Jacob Pal recognized that
physostigmine could be used to antagonize the effects of
curare. In 1912, Arthur Lawen demonstrated the use of curare
during surgery, but this potential was not realized as the finding
was published in German. In 1914, Henry Hallett Dale described the action of acetylcholine. In 1935, Harold King
isolated D-tubocurarine and described its structure, while in
1936 Dale revealed the role of acetylcholine in neuromuscular
transmission and the mechanism of action for curare. In 1940,
Abram Elting Bennett revealed that curare could be used to
reduce trauma during metrazol-induced convulsive therapy for
spastic disorders in children. In 1942, Harold Griffith and Enid
Johnson used curare to augment general anesthesia when
performing an appendectomy. Curare was used surgically until
the development of safer synthetic neuromuscular blocking
analogues such as Pancuronium (in 1964), Vecuronium
(in 1979), Mivacurium (in 1993), and Rocuronium (in 1994).
-
Physical properties
Appearance: white or slightly yellow crystalline powder. Solubility: it can be dissolved 50?mg/ml (22?°C) in water; easily soluble in methanol and ethanol; insoluble
in ether, pyridine, chloroform, benzene, and acetone; and dissolved in sodium
hydroxide solution. Specific optical rotation: +210 to +224°. Melting point: anhydrous 274–275?°C (decomposition)
-
Uses
Neuromuscular blocking agent. Historically, curare was first used as a paralyzing arrow/dart
poison by indigenous South Americans. Later, curare was used
as a muscle relaxant during surgery. Previously, to enable deep
surgery, increased relaxation could only be achieved by higher
and hence riskier quantities of general anesthetic. Being able to
control the degree of muscle relaxation independently of the
depth of sedation greatly improves survival, although bringing
an associated risk of awareness while anesthetized.
-
Clinical Use
The drug known for the muscle relaxants is mainly used for abdominal surgery and
was once used for the treatment of tremor paralysis, tetanus, rabies, poison, and so
on. For adults, the amount of one intravenous injection is 6–9?mg and can increase
to 3–4.5?mg if necessary (the amount should be reduced to 1/3?in ether anesthesia).
The action lasts for 20–40? min. The injection can be repeated according to the
length of the operation time and muscle relaxation needs, and the dose is half of the
first. For electrical shock, a dose of 0.165?mg/kg every time was administrated in
30–90?s. For diagnosis of myasthenia gravis, a dose of 0.004–0.033?mg/kg everytime was used. However, attention must be paid that the drug can lead to the risk of
paralysis of the respiratory muscles; emergency medicine and equipment must be
prepared before. Oxygen supply, endotracheal intubation, and artificial respiration
or injection of neostigmine at the same time (or phenolic ammonium chloride) can
be carried out to counteract breathing stopping. It is contraindicated for the patients
with myasthenia gravis. In addition, depolarizing muscle relaxants such as succinylcholine antagonizes non-depolarized muscle relaxant tubocurarine, and the clinical
combination should be avoided.