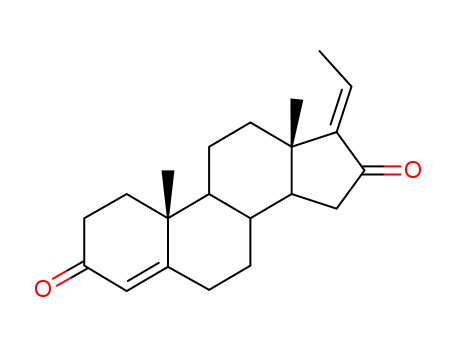
- Chemical Name:E-Guggulsterone
- CAS No.:39025-24-6
- Deprecated CAS:137682-27-0
- Molecular Formula:C21H28O2
- Molecular Weight:312.452
- Hs Code.:
- UNII:9B259YE66O
- DSSTox Substance ID:DTXSID6033538
- Wikipedia:Guggulsterone
- Wikidata:Q10910387
- Pharos Ligand ID:L818G9DRGCV3
- Metabolomics Workbench ID:133669
- ChEMBL ID:CHEMBL402063
- Mol file:39025-24-6.mol
Synonyms:(E)-guggulsterone;(Z)-guggulsterone;guggulsterone;pregna-4,17-diene-3,16-dione;pregna-4,17-diene-3,16-dione, (17E)-isomer;pregna-4,17-diene-3,16-dione, (17Z)-isomer