Chemical Property of Selanylidenestrontium
Chemical Property:
- Appearance/Colour:crystalline solid
- Melting Point:1600 °C(lit.)
- PSA:0.00000
- Density:4.540
- LogP:-0.26830
- Hydrogen Bond Donor Count:0
- Hydrogen Bond Acceptor Count:0
- Rotatable Bond Count:0
- Exact Mass:167.82213
- Heavy Atom Count:2
- Complexity:2
- Purity/Quality:
-
99% *data from raw suppliers
STRONTIUM SELENIDE 95.00% *data from reagent suppliers
Safty Information:
- Pictogram(s):
 T,
T, N
N
- Hazard Codes:T,N
- Statements:
23/25-33-50/53
- Safety Statements:
20/21-28-45-60-61
- MSDS Files:
-
SDS file from LookChem
Total 1 MSDS from other Authors
Useful:
- Canonical SMILES:[Se]=[Sr]
-
Description
Strontium selenide has the molecular formula of SrSe and the molecular weight of 166.5852 g/mol. It is a white cubic crystal whose CAS number is 1315-07-7. Its melting point is 1605°C and its density is 4.54 g/cm3. It reacts with water and so cannot be prepared by an aqueous precipitation method. It can be prepared by the reaction of the oxide with Se metal: 2SrO+ Se+ Heat ? SrSe+ SeO2 It can also be prepared by reducing the selenate with hydrogen gas: SrSeO4 + 4H2 + heat ? SrSe+ 4H2O Strontium selenide and barium selenide both have a simple cubic structure, with cube edges equal to 3.117? and 3.308 ?, respectively. In the case of SrSe, the ions at the corners are Sr++ and Se--。
-
Physical properties
Phosphors can be categorized in accordance with
their behavior as fluorescent, phosphorescent or stimulable.
As used in this disclosure, such categories
should be understood as based upon the predominant
behavior of the phosphor at about room temperature,
i.e at about 20°C.
? A “stimulable” phosphor is one that, at room
temperature, stores energy absorbed upon exposure
to excitation by electromagnetic radiation and releases
the predominant portion of the stored energy upon
exposure to stimulating electromagnetic radiation.
? A phosphorescent phosphor at room temperature will
store absorbed energy for an appreciable time but will
release the predominant portion of the stored energy
spontaneously.
? A fluorescent phosphor will release the predominant
portion of the absorbed energy as emission radiant
energy substantially simultaneously with exposure to
the exciting radiant energy.
-
Uses
The major usage of SrSe has been as an infrared stimulable
phosphor. Photoluminescent materials useful for
detection of infrared light are prepared using a base material, such as SrSe. Up to three dopants, Sm3+,
cerium, Ce3+
and Eu2+
compounds are used for the
purpose of providing electron traps. The base material
can be a combination of alkaline earth metal selenides
such as strontium selenide, calcium selenide, or
a mixture of these. Luminescence is a long known
phenomenon of nature reaching back very far in history.
Recorded observations reach back to the last century.
Seeback and Becquerel observed momentary visible
afterglow in certain materials.


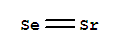
 T,
T, N
N


