Chemical Property of vanadium (V) oxide
Chemical Property:
- Appearance/Colour:yellow to rust-brown or orange crystals or powder
- Vapor Pressure:8 mm Hg ( 20 °C)
- Melting Point:690 °C
- Boiling Point:1750 °C
- Flash Point:1750oC
- PSA:77.51000
- Density:3.357 g/cm3
- LogP:-0.54360
- Storage Temp.:Poison room
- Solubility.:H2O: soluble
- Water Solubility.:1 g/125 mL
- Hydrogen Bond Donor Count:0
- Hydrogen Bond Acceptor Count:5
- Rotatable Bond Count:0
- Exact Mass:181.862488
- Heavy Atom Count:7
- Complexity:0
- Purity/Quality:
-
99.9% *data from raw suppliers
Vanadium(V) oxide *data from reagent suppliers
Safty Information:
- Pictogram(s):
 Xi,
Xi, N,
N, T
T
- Hazard Codes:Xi,N,T,C,T+
- Statements:
36/38-68-63-51/53-48/23-37-20/22-41-48/20-34-40-26/27/28
- Safety Statements:
26-61-45-38-36/37-36-36/37/39-23
- MSDS Files:
-
SDS file from LookChem
Useful:
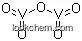
- Canonical SMILES:[O-2].[O-2].[O-2].[O-2].[O-2].[V].[V]
-
Physical properties
Brown-yellow orthorhombic crystals; density 3.35 g/cm3; melts at 670°C; decomposes at 1,800°C; slightly soluble in water, 0.8g/100 mL at 20°C; soluble in concentrated acids forming an orange-yellow solution; soluble in alkalies forming vanadates.
-
Uses
Vanadium(V) oxide is the oxide form of vanadium. As an important vanadium compound, it is used as the major precursor to alloys of vanadium and is widely used as industrial catalyst. It can be used for the production of ferrovanadium and sulfuric acid. It can also catalyze the oxidation reaction of some anhydrides. It can also be used as a detector material in bolometer arrays for thermal imaging because of its thermal resistance. Vanadium(V) oxide is also a kind of highly selective and stable ethanol sensor materials. Moreover, the microspheres of vanadium(V) oxide formed during the synthesis of vanadium(V) oxide nanorods in a mediated polyol process exhibit excellent electrochemical properties when used as the cathode material in a lithium-ion battery.
Other applications are in making yellow glass; as a depolarizer; as a developer in photography; inhibiting UV transmission in glass;and coloring ceram; as a mordant for dyeing and printing fabrics. As catalyst in the oxidation of SO2 to SO3, alcohol to acetaldehyde, etc.; for the manufacture of yellow glass; inhibiting ultraviolet light transmission in glass; depolarizer; as developer in photography; in form of ammonium vanadate as mordant in dyeing and printing fabrics and in manufacture of aniline black. Vanadium pentoxide (V2O5) is a reddish-yellow powder extracted from minerals using
strong acids or alkalies. In addition to being used as a catalyst for many organic chemical
reactions, it is used in photography and in UV-protected windowpanes and to color ceramics
and dye cloth. In the production of high-strength
steel alloys; catalyst in oxidation reactions; in
pesticides; in dyes and inks. Vanadium(V) oxide is used in Vacuum deposition, catalysts for conversion of toluene to benzonitrile or propylene to acrylonitrile, as a detector material in bolometers and microbolometer arrays for thermal imaging.
-
Description
Vanadium pentoxide is a yellow to red colour solid and is odourless. Vanadium pentoxide
dust is the particulate form of a non-combustible, odourless, yellow-orange or dark grey
crystalline solid.
On decomposition by heating, vanadium pentoxide produces toxic fumes. Vanadium
is widely distributed in the Earth’s crust in a wide range of minerals and in fossil fuels.
Vanadium pentoxide, the major commercial product of vanadium, is mainly used in the
production of alloys with iron and aluminium. It is also used as an oxidation catalyst in
the chemical industry and in a variety of minor applications.


 Xi,
Xi, N,
N, T
T


