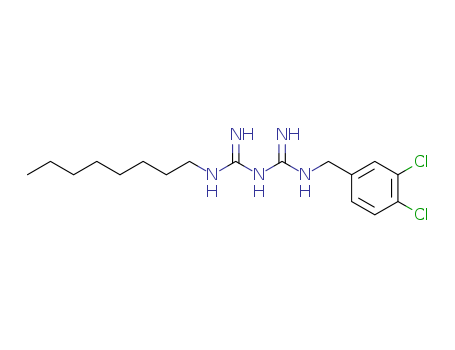
- Chemical Name:Olanexidine
- CAS No.:146510-36-3
- Molecular Formula:C17H27Cl2N5
- Molecular Weight:372.341
- Hs Code.:
- UNII:92C2328G7P
- DSSTox Substance ID:DTXSID601318290
- Nikkaji Number:J918.107E
- Wikidata:Q21099133
- NCI Thesaurus Code:C90987
- ChEMBL ID:CHEMBL1190262
- Mol file:146510-36-3.mol
Synonyms:1-(3,4-dichlorobenzyl)-5-octylbiguanide;1-(3,4-dichlorobenzyl)-5-octylbiguanide, monohydrochloride;olanexidine;OPB-2045