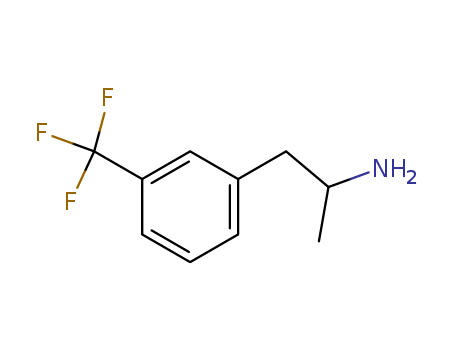
- Chemical Name:Norfenfluramine
- CAS No.:1886-26-6
- Molecular Formula:C10H12F3N
- Molecular Weight:203.207
- Hs Code.:2921499090
- NSC Number:43036
- UNII:037A9J3PSW
- DSSTox Substance ID:DTXSID60904717
- Nikkaji Number:J50.067D
- Wikipedia:Norfenfluramine
- Wikidata:Q3069482
- Pharos Ligand ID:B5JX9WZ82R6K
- ChEMBL ID:CHEMBL1979333
- Mol file:1886-26-6.mol
Synonyms:Dexnorfenfluramine;Nordexfenfluramine;Norfenfluramine