10.1055/s-2005-871564
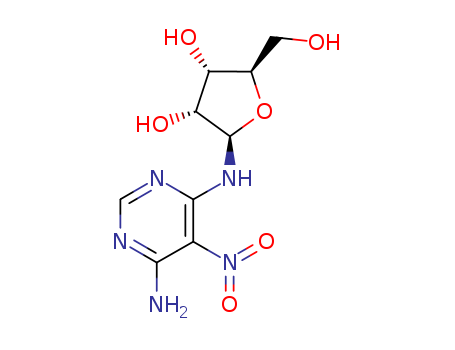
The research aims to develop an efficient synthesis method for clitocine, a compound with significant biological activity, including insecticidal properties and cytostatic effects on leukemia cell lines. The study revisits Kini's previous work and introduces a novel synthetic pathway involving a 1,3-N (endo) to N (exo) migration as a key transformation. The researchers used p-chlorobenzoyl (PCB) as a protecting group to facilitate the solidification of intermediates and minimize epimerization at the final deprotection stage. Key chemicals used in the synthesis include 4,6-diamino-5-nitropyrimidine, 1-O-acetyl-2,3,5-tri-O-benzoyl-D-ribofuranose, and silica gel for isomerization. The study concludes that the endo-product formed during glycosylation is kinetically favored but isomerizes to the thermodynamically stable exo-product under acid catalysis. The incorporation of the PCB group significantly improves the isolation and purification of intermediates. The final deprotection step using methanol and sodium methoxide yields highly pure clitocine without any column separation, demonstrating a more efficient and practical synthesis route compared to previous methods.