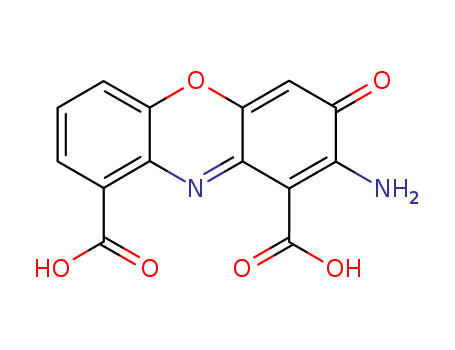
Chemical Property of Cinnabarinic acid
Chemical Property:
- Vapor Pressure:2.36E-12mmHg at 25°C
- Melting Point:>300°C
- Boiling Point:536.8°C at 760 mmHg
- Flash Point:278.4°C
- PSA:143.72000
- Density:1.79
- LogP:1.85260
- Storage Temp.:Refrigerator
- Solubility.:DMSO: ≥4mg/mL
- XLogP3:0.8
- Hydrogen Bond Donor Count:3
- Hydrogen Bond Acceptor Count:8
- Rotatable Bond Count:2
- Exact Mass:300.03823598
- Heavy Atom Count:22
- Complexity:675
- Purity/Quality:
-
97% *data from raw suppliers
Cinnabarinic acid *data from reagent suppliers
Safty Information:
- Pictogram(s):
- Hazard Codes:
- MSDS Files:
-
SDS file from LookChem
Useful:
- Canonical SMILES:C1=CC(=C2C(=C1)OC3=CC(=O)C(=C(C3=N2)C(=O)O)N)C(=O)O
-
Description
Cinnabarinic acid is a phenoxazinone produced by the oxidative dimerization of 3-hydroxyanthranilic acid (3-HAA) as part of the metabolism of tryptophan in the kynurenic pathway. It acts as a partial receptor agonist of the metabotropic glutamate receptor 4 (mGlu4), effective at 100 μM, with no activity at other mGlu receptor subtypes. 3-HAA does not affect mGlu receptors, including mGlu4. Cinnabarinic acid induces apoptosis of T cells at 300-500 μM, a potency some ten times that of 3-HAA.
-
Uses
A natural phenoxazinone deriv
Cin
nabarinic acid strongly induces apoptosis in thymocytes through the generation of reactive oxygen species and the induction of caspase. A natural phenoxazinone derivative, Cinnabarinic acid is obtained in vitro with aid of laccase by oxidative dimerization of 3-Hydroxyanthranilic acid (a metabolite of the amino acid Trytophan). Cinnabarinic acid strongly induces apoptosis in thymocytes through the generation of reactive oxygen species and the induction of caspase. Cinnabarinic acid may be used in studies of the functions of components of the kynurenine metabolic pathway. It may be used to study it role as a metabotropic glutamate receptor (mGlu4R-specific) agonist.