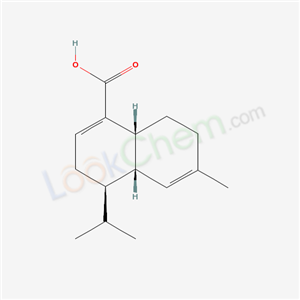
- Chemical Name:Sclerosporin
- CAS No.:66419-03-2
- Molecular Formula:C15H22O2
- Molecular Weight:234.334
- Hs Code.:
- UNII:DR07Q5J492
- DSSTox Substance ID:DTXSID70331818
- Nikkaji Number:J2.771.099I
- Wikidata:Q27108254
- ChEMBL ID:CHEMBL1087671
Synonyms:Sclerosporin;(+)-Sclerosporin;66419-03-2;UNII-DR07Q5J492;DR07Q5J492;CHEBI:9054;1-Naphthalenecarboxylic acid, 3,4,4a,7,8,8a-hexahydro-6-methyl-4-(1-methylethyl)-, (4R,4aS,8aR)-;(4R,4aS,8aR)-6-methyl-4-propan-2-yl-3,4,4a,7,8,8a-hexahydronaphthalene-1-carboxylic acid;1-Naphthalenecarboxylic acid, 3,4,4a,7,8,8a-hexahydro-6-methyl-4-(1-methylethyl)-, (4R-(4alpha,4aalpha,8aalpha))-;C09721;CHEMBL1087671;DTXSID70331818;Q27108254;1-NAPHTHALENECARBOXYLIC ACID, 3,4,4A,7,8,8A-HEXAHYDRO-6-METHYL-4-(1-METHYLETHYL)-, (4R-(4.ALPHA.,4A.ALPHA.,8A.ALPHA.))-