10.1039/c0cc02865g
The study presents an in situ coupled oxidation cycle that enables catalytic oxidation of substrates using catalytic amounts of o-chloranil and novel, reusable polymer-immobilized platinum nanocluster catalysts, with molecular oxygen serving as the terminal oxidant. The chemicals used in the study include o-chloranil, a quinone with high oxidation potential, and platinum nanocluster catalysts, which are immobilized on a polymer matrix. These catalysts were designed to overcome the challenges associated with the stoichiometric use of quinones in organic synthesis, such as difficulty in removing by-products and high costs. The purpose of these chemicals was to facilitate a more environmentally friendly and economically viable oxidation process, where molecular oxygen, being abundant and producing water as the only by-product, was utilized as the ideal oxidizer. The study demonstrated the effectiveness of this system in the oxidation of dihydropyridines to pyridines, among other reactions, and showed that the platinum catalysts could be reused multiple times without significant loss of activity.
10.1016/S0040-4039(00)94524-6
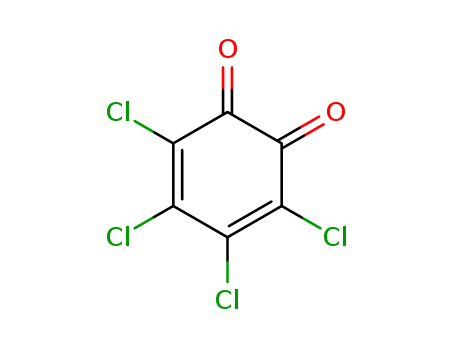
The research investigated the photoreactions of certain triarylfurobenzodioxin derivatives, focusing on how the type and position of substituents on the 9a-aryl group, as well as the wavelength of light used, affect the reaction outcomes. The study synthesized these derivatives by reacting 1,2,4-triarylbuten-1,4-diones with concentrated hydroiodic acid to form phenylated furans, which were then reacted with tetrachloro-1,2-benzoquinone to produce the desired dihydrobenzodioxins. The researchers found that the presence of electron-withdrawing groups at the para-position of the 9a-aryl group altered the reaction pathway, leading to a competition between the cis-stilbene cyclisation-elimination process and a retro-Diels-Alder reaction.


 Xn
Xn N
N




