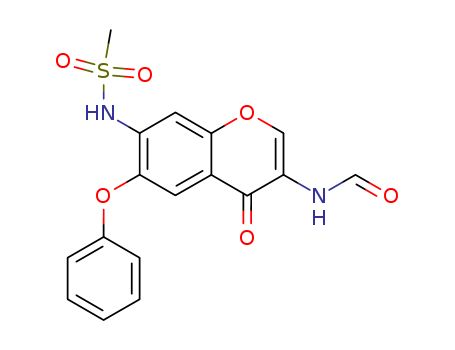
Chemical Property of Iguratimod
Chemical Property:
- Appearance/Colour:white powder
- Vapor Pressure:1.8E-13mmHg at 25°C
- Melting Point:238.0 to 242.0 °C
- Refractive Index:1.673
- Boiling Point:580.6 °C at 760 mmHg
- PKA:5.58±0.20(Predicted)
- Flash Point:304.9 °C
- PSA:123.09000
- Density:1.52 g/cm3
- LogP:4.38780
- Storage Temp.:under inert gas (nitrogen or Argon) at 2-8°C
- Solubility.:DMSO (Slightly)
- XLogP3:1.4
- Hydrogen Bond Donor Count:2
- Hydrogen Bond Acceptor Count:7
- Rotatable Bond Count:5
- Exact Mass:374.05725734
- Heavy Atom Count:26
- Complexity:665
- Purity/Quality:
-
99% *data from raw suppliers
Iguratimod *data from reagent suppliers
Safty Information:
- Pictogram(s):
- Hazard Codes:
- MSDS Files:
-
SDS file from LookChem
Useful:
- Canonical SMILES:CS(=O)(=O)NC1=C(C=C2C(=C1)OC=C(C2=O)NC=O)OC3=CC=CC=C3
- Recent ClinicalTrials:The Clinical Efficacy of Immunomodulators in RA Patients
- Recent NIPH Clinical Trials:Effect of tapering methotrexate in rheumatoid arthritis patients after add-on iguratimod therapy.
-
Description
In August 2011, China’s State FDA approved Simcere Pharmaceutical
Group’s new drug application for iguratimod (T-614), a disease modifying
anti-rheumatic drug (DMARD) for the treatment of rheumatoid arthritis
(RA). Preclinical in vivo studies indicated that iguratimod was effective
in an established adjuvant-induced arthritis model (ED40=3.6 mg/kg)
in rats and also efficacious in a type II collagen-induced arthritis model in DBA/1J mice at 30 mg and 100 mg/kg.
-
Uses
Iguratimod acts as an anti-inflammatory agent, used primarily in the treatment of rheumatoid arthritis.
-
Clinical Use
Iguratimod, which was discovered by Toyama Pharmaceuticals and jointly co-developed with Eisai in
Japan, was approved by the PMDA (Pharmaceuticals and Medical Devices Agency) of Japan on June 29,
2012 for the treatment of rheumatoid arthritis. This drug was also independently developed by
Simcere Pharmaceutical Group and is marked as Iremod? in China. The drug exhibited inhibitory
effects on granuloma inflammation, and was shown to be efficacious for the prevention of joint
destruction in adjuvant arthritis.