10.1021/jm00313a025
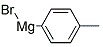
The research focuses on the chemical synthesis and analysis of various purine derivatives, specifically 6-(alkylthio)-9-(hydroxymethyl)-SH-purines. The study investigates the hydroxymethylation of purine-6(1H)-thione and its derivatives, revealing that substitution occurs at the 9 position of the purine ring. Key chemicals involved include ethyl acetate, ligroin, benzene, ethyl alcohol, and petroleum ether, used as solvents in the synthesis and analysis processes. The researchers also utilized aqueous formaldehyde and sodium carbonate for hydroxymethylation reactions. Additionally, the study examines the infrared and ultraviolet spectra of these compounds, as well as their stability and reactivity with isocyanates. One of the synthesized compounds was screened for antitumor activity, showing promising results in certain leukemia models. The research provides valuable insights into the structure and potential applications of these purine derivatives.