10.1039/b717437c
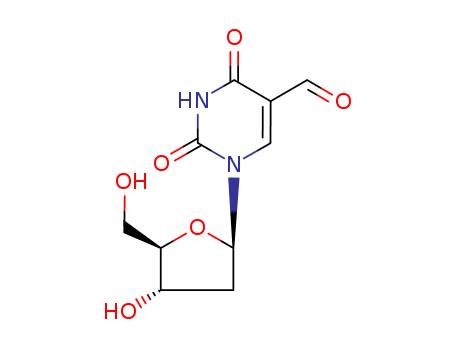
The research investigates the one-electron oxidation of DNA duplex oligomers that do not contain guanine, focusing on the reactions at thymine bases. The purpose is to understand the mechanisms and products of oxidation in DNA sequences lacking guanine, which is typically the most reactive base in DNA oxidation. The study uses anthraquinone (AQ) as a photosensitizer linked to DNA oligomers to generate radical cations upon UVA irradiation. The key findings are that thymine, despite having a higher oxidation potential than adenine, is the primary site of oxidation reactions, leading to products such as thymidine glycols, 5-(hydroxymethyl)-2'-deoxyuridine, and 5-formyl-2'-deoxyuridine. 5-Hydroxymethyl-2'-deoxyuridine (5-HMdUrd) is formed through the reaction of the thymine radical cation with molecular oxygen (O2) after the initial deprotonation of the thymine methyl group. This process involves the formation of a transient 5-(2'-deoxyuridinyl)methyl radical, which is subsequently trapped by O2. 5-Formyl-2'-deoxyuridine (5-FormdUrd) is another product formed from the reaction of the thymine radical cation. Similar to 5-HMdUrd, its formation involves the initial deprotonation of the thymine methyl group, followed by reaction with molecular oxygen (O2). The research concludes that the reactivity of the thymine radical cation, rather than its stability, determines the oxidation products. The study also proposes a mechanism involving proton loss from the thymine methyl group or addition of H2O/O2 across the thymine double bond, which can initiate tandem reactions converting both thymines in a TT step to oxidation products. This work has implications for understanding oxidative damage in genomic DNA, particularly in sequences with few guanines.