Chemical Property of Mercuric sulfide
Chemical Property:
- Appearance/Colour:Odorless red or black solid
- Melting Point:583.5 °C(lit.)
- Boiling Point:584 °C
- PSA:32.09000
- Density:8.1 g/mL at 25 °C
- LogP:0.64570
- Hydrogen Bond Donor Count:0
- Hydrogen Bond Acceptor Count:1
- Rotatable Bond Count:0
- Exact Mass:233.942715
- Heavy Atom Count:2
- Complexity:2
- Transport DOT Label:Poison
- Purity/Quality:
-
99% *data from raw suppliers
Safty Information:
- Pictogram(s):
 T,
T, N
N
- Hazard Codes:
 N-T:;
N-T:;
- Statements:
50/53-36-33-23/24/25:;
- Safety Statements:
61-60-45-36/37/39-28A-26:;
- MSDS Files:
-
SDS file from LookChem
Useful:
- Chemical Classes:Metals -> Mercury Compounds, Inorganic
- Canonical SMILES:S=[Hg]
-
Use Description
Mercury(II) sulfide, a chemical compound with the formula HgS, finds applications in various fields. In the field of electronics, it has been used in the manufacture of electrical switches and rectifiers due to its semiconductor properties. Its ability to conduct electricity under specific conditions makes it valuable in electronic components. Additionally, in the field of cosmetics and pigments, mercury(II) sulfide, also known as vermilion, has been historically used as a bright red pigment in paints, dyes, and cosmetics. Its vibrant color and stability have made it popular in artistic and cosmetic applications. However, it's important to note that due to the toxicity of mercury, its use in cosmetics has significantly declined over the years. Overall, its role as a semiconductor in electronics and as a pigment in cosmetics highlights its significance in these respective fields, although its use in cosmetics has become limited due to health concerns.

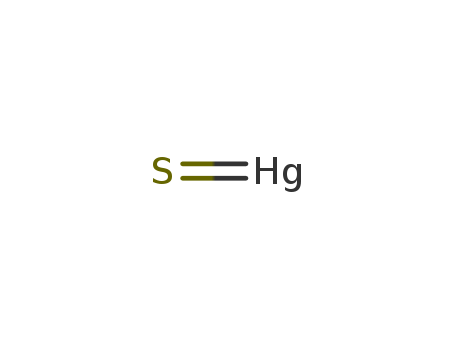


 T,
T, N
N
 N-T:;
N-T:;



