Chemical Property of Nickel(III) oxide
Chemical Property:
- Appearance/Colour:solid
- Melting Point:decomposes at ~600℃ [CRC10]
- PSA:0.00000
- Density:4.83 g/cm3
- LogP:-0.11880
- Water Solubility.:insoluble H2O; very slightly soluble cold acid; dissolves in hot HCl releasing Cl2; dissolves in hot H2SO4 or HNO3 evolving O2 [MER06]
- Hydrogen Bond Donor Count:0
- Hydrogen Bond Acceptor Count:3
- Rotatable Bond Count:0
- Exact Mass:163.855427
- Heavy Atom Count:5
- Complexity:0
- Purity/Quality:
-
99% *data from raw suppliers
Nickel oxide *data from reagent suppliers
Safty Information:
- Pictogram(s):
 T
T
- Hazard Codes:T
- Statements:
49-43-53
- Safety Statements:
53-45-61
- MSDS Files:
-
SDS file from LookChem
Total 1 MSDS from other Authors
Useful:
- Chemical Classes:Metals -> Nickel Compounds, Inorganic
- Canonical SMILES:[O-2].[O-2].[O-2].[Ni+3].[Ni+3]
-
Uses
Storage batteries. Nickel oxide (NiO) is produced from nickel minerals to form nickel oxide when heated to
400°C, which is then reduced at a temperature of 600°C, resulting in the formation of nickel
oxide. It is used as electrodes in fuel cells. Nickel oxide is used in the ceramic industry for making frit, ferrites, and coloring porcelain. The oxide in sinter form is used in the production of nickel-steel alloys. It supplies oxygen to the melt for removal of carbon as carbon dioxide. Some other important uses of nickel oxide include preparation of many nickel salts, specialty chemicals, and nickel catalysts. It also is used as an electrode in fuel cells. ▼▲
Industry
Application
Role/benefit
Chemical manufacture
Manufacture of nickel salts and other nickle compounds
Source of nickel
Manufacture of metallic nickel
Manufacture of nickel steel alloys
Ceramic pigments
Coloring of ceramics and glass
NiO-based black ceramic pigments
Electrical cell
Nickel-iron battery
Active material of negative electrode
Ni-Cd rechargeable batteries
Ni-MH battery
Active material of positive electrode
Tandem solar cell
Photocathodes material
Electronic
Electrochromic devices
Anodic electrochromic material
Others
Hydrogenation reaction
Catalyst

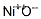

 T
T


