Chemical Property of Oxo(oxocobaltiooxy)cobalt
Chemical Property:
- Appearance/Colour:black powder
- Melting Point:895 °C (dec.)(lit.)
- PSA:43.37000
- Density:6.11 g/mL at 25 °C(lit.)
- LogP:-0.30600
- Storage Temp.:Inert atmosphere,Room Temperature
- Water Solubility.:insoluble H2O; soluble conc acids [HAW93]
- Hydrogen Bond Donor Count:0
- Hydrogen Bond Acceptor Count:3
- Rotatable Bond Count:0
- Exact Mass:165.851131
- Heavy Atom Count:5
- Complexity:0
- Purity/Quality:
-
99% *data from raw suppliers
Cobalt(II,III) oxide nanopowder, <50?nm particle size (TEM), 99.5% trace metals basis *data from reagent suppliers
Safty Information:
- Pictogram(s):
 Xn
Xn
- Hazard Codes:Xn
- Statements:
36/37/38-40-43-22
- Safety Statements:
26-36/37/39-36/37
- MSDS Files:
-
SDS file from LookChem
Useful:
- Canonical SMILES:[O-2].[O-2].[O-2].[Co+3].[Co+3]
-
Uses
COBALT(III) OXIDE BLACK is used as a pigment; for glazing porcelain and pottery; and for coloring enamels. Cobaltic oxide (Co2O3) is also known as cobalt oxide or cobalt black. It is dark gray to black
and is used in pigments, ceramic glazes, and semiconductors. Pigment, coloring enamels, glazing pottery.
-
Description
Neither the oxide Co2O3 nor the hydroxide Co(OH)3 is definitely established. The oxidation of cobalt(II) hydroxide in aqueous suspension with, for example, peroxides, or the destruction of a cobalt(III) complex with alkali gives a brown or black powder Co2O3.aq which upon drying at 150° yields the monohydrate Co2O3.H2O; this is probably best formulated as CoO(OH). When heated further in attempts to dehydrate it, the "monohydrate" begins to evolve oxygen (as well as water) at 300° with the formation of black Co3O4. When heated to 100° in air cobalt(II) hydroxide is converted to dark brown CoO(OH).
-
Physical properties
Grayish black powder; density 5.18 g/cm3; decomposes at 895°C; insoluble in water; soluble in concentrated mineral acids.

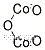

 Xn
Xn


