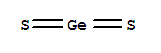Base Information
- Chemical Name:Germanium disulfide
- CAS No.:12025-34-2
- Deprecated CAS:145114-12-1,145114-13-2,145114-13-2
- Molecular Formula:GeS2
- Molecular Weight:136.77
- Hs Code.:
- European Community (EC) Number:234-705-1
- DSSTox Substance ID:DTXSID2065177
- Nikkaji Number:J43.815D
- Wikipedia:Germanium_disulfide,Germanium(IV) sulfide
- Wikidata:Q2420582
- Mol file:12025-34-2.mol
Synonyms:GERMANIUM DISULFIDE;12025-34-2;bis(sulfanylidene)germane;Germanium disulphide;Germanium sulfide (GeS2);Germanium(IV) sulfide;Germanium sulfide(GeS2);EINECS 234-705-1;dithioxogermane;GeS2;Germanic sulfide;GERMANIUMDISULFIDE;Ge-S2;DTXSID2065177;Germanium Disulfide (GeS2) Crystals;MFCD00016122;AKOS015909186;CS-0435365;FT-0694973;J-004291;Q2420582